Serviços Personalizados
Journal
artigo
Indicadores
Compartilhar
Estudos de Psicologia (Natal)
versão impressa ISSN 1413-294Xversão On-line ISSN 1678-4669
Estud. psicol. (Natal) vol.25 no.1 Natal ene./mar. 2020
https://doi.org/10.22491/1678-4669.20200003
10.22491/1678–4669.20200003
PSYCHOBIOLOGY E COGNITIVE PSYCHOLOGY
Executive functions: neuropsychological assessment in women with breast cancer
Funções executivas: avaliação neuropsicológica em mulheres com câncer de mama
Funciones ejecutivas: evaluación neuropsicológica en mujeres con cáncer de mama
Camila Vasconcelos Carnaúba LimaI; Rosa Maria Martins de AlmeidaII; Raner Miguel Ferreira PóvoaIII
IUniversidade Tiradentes
IIUniversidade Federal do Rio Grande do Sul
IIIUniversidade Federal de Alagoas
ABSTRACT
Cognitive changes may occur during chemotherapy to treat cancer and, therefore, it is necessary to identify them since they represent neurological complications with a high rate of improvement when treated. This research evaluated executive functions in women with breast cancer undergoing chemotherapy. Were examined 171 women divided into three groups contrasted with the use of neuropsychological tests. Performance losses were found in the Digit Span both direct (p < 0.001) and indirect sequence task (p < 0.001), Victoria Stroop Test (p = 0.017) and BIS–11 (p = 0.004). It is concluded that there were impairments in executive functions related to working memory, focused attention, inhibitory control and general impulsivity in the group of women with breast cancer who received chemotherapy. Studies have corroborated these results, but there are still gaps in the literature. Discussion on the topic is encouraged with the goal of building intervention strategies.
Keywords: executive dysfunction; chemotherapy; breast carcinoma.
RESUMO
Alterações cognitivas podem ocorrer durante a quimioterapia no tratamento de câncer e, portanto, faz–se necessário a identificação destas, já que representam complicações neurológicas com alto índice de melhora quando tratadas. Essa pesquisa avaliou as funções executivas em mulheres com câncer de mama submetidas à quimioterapia. Foram examinadas 171 mulheres divididas em três grupos contrastados com o uso de testes neuropsicológicos. Foram encontrados prejuízos de desempenho nas tarefas de sequência direta do Digit Span (p = 0,000), de sequência indireta do mesmo (p = 0,000), Victória Stroop Test (p = 0,017) e BIS–11 geral (p = 0,004). Conclui–se que houve prejuízos das funções executivas relativas à memória de trabalho, atenção focada, controle inibitório e impulsividade geral no grupo de mulheres com câncer de mama que receberam quimioterapia. Estudos tem corroborado com tais resultados, mas ainda há lacunas na literatura. Espera–se fomentar discussão sobre o tema e contribuir na construção de estratégias de intervenção.
Palavras–chave: disfunção executiva; quimioterapia; carcinoma de mama.
RESUMEN
Los cambios cognitivos pueden ser causados durante la quimioterapia en el tratamiento del cáncer y, por lo tanto, es necesario identificarlos, ya que representan complicaciones neurológicas con una alta tasa de mejora cuando se tratan. Esta investigación evaluó las funciones ejecutivas en mujeres con cáncer de seno sometidas a quimioterapia. Se examinaron 171 mujeres, divididas en tres grupos, en contraste con el uso de pruebas neuropsicológicas. Se encontraron pérdidas de rendimiento en las tareas de secuencia directa de Digit Span (p = 0.000), secuencia indirecta de la misma (p = 0.000), Victória Stroop Test (p = 0.017) y general BIS–11 (p = 0.004). Se concluye que hubo deficiencias en las funciones ejecutivas relacionadas con la memoria de trabajo, atención focalizada, control inhibitorio e impulsividad general en el grupo de mujeres con cáncer de seno que recibieron quimioterapia. Los estudios han corroborado estos resultados, pero todavía hay lagunas en la literatura. Se espera que fomente la discusión sobre el tema y contribuya a la construcción de estrategias de intervención.
Palabras clave: disfunción ejecutiva; quimioterapia; carcinoma de mama.
Cancer is a pathology caused by the uncontrollable growth of abnormal cells in the body which can invade adjacent tissues and spread to other organs through the bloodstream and the lymphatic system (Instituto Nacional de Câncer [INCA], 2012). This growth forms a malignant tumor characterized in an atypical manner, with formation of anaplastic cells (different from those of normal tissue), which develop quickly with abnormal and numerous mitoses (INCA, 2016).
Currently, the breast is the primary affected cancer location in woman in the world (24.2%). Incidence and mortality rates due to breast cancer predominate in different countries regardless of the Human Development Index. In Brazil, in 2017, there were 16,724 deaths from breast cancer. For the three–year period from 2020 to 2022, there will be an estimated 66,280 new (29.7%) cases of breast cancer in women, the number one type in this public (except for non–melanoma skin cancer). This reflects an estimated risk of 61.61 new cases per 100,000 women. Furthermore, this is the type of cancer most suffered by women in the Northeast region, being the second region with the highest incidence in the country (INCA, 2019).
Treatment through chemotherapy has been the most promising alternative for fighting cancer, capable of destroying small foci of tumor growth that spread throughout the body and cannot be accessed by other routes (Jemal, Vineis, Bray, Torre, & Forman, 2014). The therapeutic protocol for breast cancer consists of combinations of drugs (chemotherapy regimens) such as anthracyclines (doxorubicin and epirubicin), taxanes (paclitaxel and docetaxel), cyclophosphamide, methotrexate, Carboplatin and 5–fluorouracil (Ordinance No. 1,008, 2015). On the other hand, chemotherapy does not only act on cancer cells it also affects other structures under normal conditions that are constantly renewed, such as bone marrow, hair and the mucosa of the digestive tract. The most well–known effects of chemotherapy are myelodepression, hair loss, nausea, vomiting and diarrhea (INCA, 2012), however, studies have shown that this type of treatment can also cause cognitive impairments, thus reducing its functionality (Chen et al., 2014; Ganz et al., 2013, Koppelmans et al., 2012; Lyon et al., 2016; Park, Bae, Jung, & Jung, 2015; Yao et al., 2016; Zheng et al., 2014).
Chemotherapy targets catechol O–methyltransferase (COMT), an important dopamine modulator, and may be associated with increased cytokines and reduced rates of estrogen and testoterone, which directly or indirectly interferes with cognitive performance. In this case, women undergoing chemotherapy are subject to abruptly experiencing a menopause, that is, anticipating their symptoms in cognitive impairment, mainly in terms of working memory (Ahles & Saykin, 2007). During and after chemotherapy, cognitive complaints have been constantly reported by women, particularly those related to attention (Piccirillo et al., 2015), memory and executive function (Ahles & Saykin, 2007; Ganz, 2013).
The executive functions refer to three central skills, according to the theoretical model presented by Adele Diamond (2013): 1) inhibition; 2) working memory; 3) cognitive flexibility. The first involves inhibitory control, whose ability is to manage attention, behavior and thoughts and / or emotions to replace a strong internal predisposition (tendency to respond on impulse or old habits) or to select more appropriate or necessary attitudes. In this way, it is possible to focus on something selectively chosen, suppressing attention to other stimuli. It also has the function of eliminating dominant mental representations, such as resisting strange thoughts or reactive interference from unwanted memories.
On the other hand, working memory involves the ability to keep information in mind and work with it, imagining what has happened before and what may happen after a given action. It is a reasoning that allows visualizing connections between apparently unrelated things and separating elements that were integrated. This implies creativity, as it involves new ways of disassembling and recombining available elements. It also influences decision making, as it allows a conceptual knowledge of such elements (in addition to mere perceptions) and their relationship with the past and future. The role of the dorsolateral cortex in maintaining this memory stands out (Diamond, 2013).
Cognitive flexibility refers to the ability to change spatial or interpersonal perspectives through the inhibition of actions, thoughts or emotions that are already part of a habit, activating the working memory to think about new possibilities in a given situation. It is necessary to change the point of view and think "outside the box". Flexibility, therefore, is the opposite of behavioral rigidity, in addition to being correlated to creativity (Diamond, 2013).
To the prefrontal cortex is assigned the role of "executive" because of its synthesizing information synthesis function from a wide range of brain systems and exercising control over behavior (Nauta, 1971). Interconnections are made with areas of external information processing (sensory, cortical and subcortical systems) and internal (limbic and mensencephalon systems involved in affection, memory and reward) and their functions have a top–down influence, that is, from cortical areas to subcortical. The damage to the prefrontal cortex in an initial examination is not very noticeable, since the patient continues to exercise his life normally, without appearing to have impairments in memory, perception and movement. However, he may have difficulties in maintaining attention on a given task, as well as behaving in an impulsive manner, being able to make decisions without assessing future consequences (Miller; Freedman; & Wallis, 2002).
Although oncology has been an area of considerable interest in research, the emphasis on cognitive aspects still needs to be further investigated on correlations with prevalence, severity, stages of treatment and survival, as well as the most affected domains (Lyon et al., 2016). There is also a need for studies that use case–control methodology (Ando–Tanabe et al., 2012) and that explore the cognitive performance of women in pre–treatment, comparing with others in post–treatment (Lyon et al., 2016). Thus, the high cancer survival rate and the prevalence of cognitive impairments justifies the need for further studies on the cognitive aspects of these people, after medical treatment, since it negatively impacts quality of life and goal planning (Piccirillo et al., 2015). Such research can point out important information aiming at the construction of interventions that can improve the daily functioning and the quality of life of these people (Kam et al., 2015).
Bearing in mind that executive functions are essential skills for social, emotional and physical health and have a great prospect of improvement when they are impaired and are rehabilitated (Diamond, 2013), this research aimed to examine the executive performance of women with breast cancer undergoing chemotherapy treatment in aspects of working memory, focused attention, inhibitory control / cognitive flexibility, impulsivity and decision making. In addition, information on age, education, anxiety and depression were analyzed in order to verify whether they have any interference or significant effect on the executive functions evaluated. For comparative purposes, the performance of women with and without a diagnosis of breast cancer who have never undergone chemotherapy was also evaluated.
Method
This is a cross–sectional survey with non–probabilistic sampling for convenience with three contrasting groups: CQ Group – women diagnosed with breast cancer undergoing chemotherapy; SQ Group – women who were diagnosed with breast cancer but had not yet undergone chemotherapy treatment; and GC Group – healthy women in the control group. The groups were composed of 55 participants in the CQ, 52 in the SQ, and, 64, in the GC.
The participants in the groups with breast cancer – CQ and SQ– had a recent history of breast cancer illness (in primary location). In the CQ group, the women had finished chemotherapy, at most 3 months before, or were in the last three treatment sessions.
All participants in this study were in the age group between 30 and 59 years old and had elementary or secondary education. Were excluded women with cognitive function deficits, previously diagnosed and with problems related to immunity (for example, multiple sclerosis, systemic lupus erythematosus), in addition to those with evidence of other cancer, or in active psychosis. In any of the groups, women who were illiterate or who had a history of head trauma or loss of consciousness were not part of the research. Were also excluded for the selection of the control group, women diagnosed with cancer or who had already undergone chemotherapy, underwent medical treatment of chronic disease, in addition to those who consumed tobacco and alcohol on a daily basis.
For this research the data from the group composed of CQ and SQ were collected in oncological clinics1 that offer diagnosis and treatment services for people with cancer, available in a state in the Northeast region of Brazil. Those in the control group, were obtained in the same clinics and other institutions, with healthy women chosen according to the aforementioned inclusion and exclusion criteria.
The research protocol was composed of four parts divided into: 1) access and invitation from the possible participant with exposure / discussion on the Free and Informed Consent Term; 2) filling in anamnesis; 3) application of instruments for neuropsychological assessment in the following order: Digit Span; Stroop Test; BIS–11; IGT; 4) application of instruments to assess levels of anxiety and depression. This protocol lasted about 50 minutes.
The following instruments were used: Digit Span (Digit Span), a subtest of the 3rd focused attention and working memory; Victoria Stroop test, according to Strauss (2006), to assess inhibitory control and cognitive flexibility; Iowa Gambling Task– IGT in Brazilian version (Malloy–Diniz et al., 2008) to evaluate decision making; Barratt's Impulsivity Scale, BIS–11 (Malloy–Diniz et al., 2010) with Cronbach's alpha (internal consistency) of 0.77 (Salvo & Castro, 2013), to assess general, attentional, motor and lack of impulsivity of planning; the Hospital Anxiety and Depression Scale (HADS) by Snaith (2003) to assess levels of anxiety (with Cronbach's alpha of 0.84) and depression (with Cronbach's alpha of 0.83), according to Marcolino and collaborators (2007).
The collected data was tabulated and analyzed statistically using the Statistical Package for the Social Sciences (SPSS 22 Chicago, Illinois), in its version for Windows. Quantitative variables were described through central tendency (mean and median) and variability / dispersion (standard deviation), while qualitative variables were described using absolute and relative frequency.
The variables referring to the components of the executive functions were related to the independent variables (chemotherapy, breast cancer without chemotherapy and healthy) and tested for difference of means using statistical hypothesis tests. In cases where the frequencies showed normal distribution, the Analysis of Variance (ANOVA) technique was used, otherwise the Kruskal–Wallis test was performed. To check which of the groups differed from each other, post hoc analysis were performed with Tukey tests. Were considered statistically significant p–values lower than 0.05 (5%).
This research was submitted to Plataforma Brasil on 04/03/2017 and approved, obtaining the Certificate of Presentation for Ethical Appreciation (CAAE) from the Ministry of Health, under the number: 62265916.3.0000.5013. These procedures are in line with the proposals of the National Health Council regarding the standards for research with human beings established in Resolution No. 466 of 12 November 2012.
Results
Table 1 shows the characterization of the research participants. In the control group (CG), there was a greater predominance in the age group between 30 to 39 years, while the other groups (CQ and SQ) predominated between 40 and 55. It is observed that the age group of the diagnosed women with cancer (both CQ and SQ groups) is closer together than when compared to those in the control group. This can be explained by the higher incidence of breast cancer in women over 35 years, especially after 50 years of age, according to data from INCA (2019).
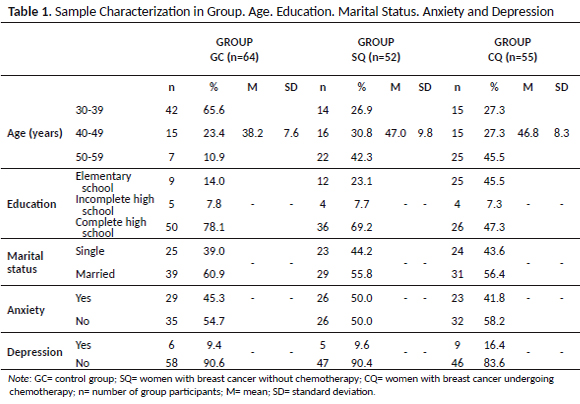
Table 2 presents a summary of the descriptive and inferential statistical values of the participants' performance in the research. Statistically significant differences were found between the means of the performance values, when the groups were compared using ANOVA, in the tests of focused attention (p < 0.001) evaluated by the direct sequence Digit Span; working memory (p < 0.001), by reverse sequence Digit Span; inhibitory control (p = 0.017), assessed by the Stroop Test; general (p = 0.004) and attentional impulsivity (0.017) and lack of planning (p = 0.004), assessed by BIS–11. The errors issued by the participants during the Stroop Test did not have a normal distribution, and, therefore, the Kruskal–Wallis test was performed in the three groups with an associated probability value of 0.018, pointing out significant differences. The motor impulsivity variables, assessed by the BIS–11 test, and decision making by the IGT, do not present significant differences between the groups.
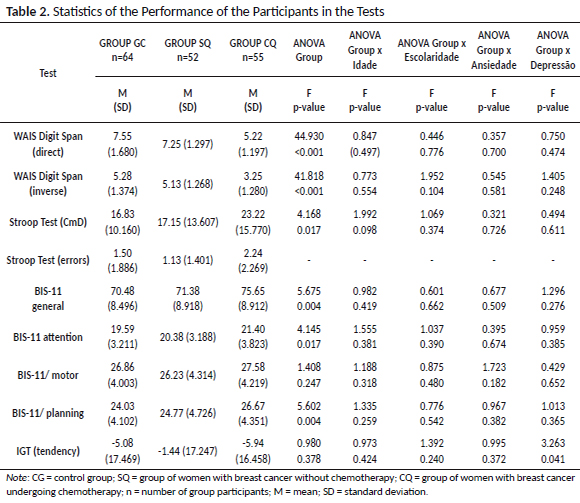
As seen, statistical tests pointed out differences in seven evaluated variables. However, post hoc analyzes indicated that the group undergoing chemotherapy (CQ) only performed differently from the others (SQ and GC) in focused attention, working memory, inhibitory control / cognitive flexibility and general impulsivity, as shown in Table 3.
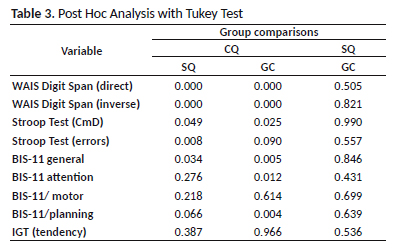
The same statistical tests were performed to verify the interference of age, education, anxiety and depression factors when individually associated with the sample groups. No significant differences were found in the average performance of executive functions for each of these checks, except for decision making associated with depression with a p–value = 0.041 – but this difference was discarded after the post hoc analysis. This indicates a low probability of these variables interfering in the performance of each group.
In summary, the results of this research suggest that chemotherapy in patients with breast cancer may be associated with cognitive deficits related to working memory, focused attention, inhibitory control / cognitive flexibility and general impulsivity.
Discussion
The present study indicated that women who received chemotherapy obtained inferior results in executive functions tasks when compared to other women (with and without cancer) who did not receive this type of treatment. The results will be discussed below based on data of corroborations and controversies found in other studies whose method was similar.
With regard to working memory, studies by Park et al. (2015), Chen et al. (2014) and Askren et al. (2014) showed a decrease in performance for women undergoing treatment. A study with neuroimaging by Dumas et al. (2013) pointed out harmful effects of chemotherapy on functional brain connectivity related to working memory, assessed by N–back task, at one month and one year after treatment. In this sense, McDonald, Conroy, Smith, Oeste and Saykin (2013) found a decrease in the frontal density of gray matter after the first month of completion of chemotherapy, especially in the frontal regions. This change was compatible with the self–reported difficulties with executive functioning and with impaired working memory performance, assessed by BRIEF–A, in addition to the increase in self–perceived symptoms of the ability to initiate problem solving or an activity. On the other hand, controversies were detected in the results of the studies by Ando–Tanabe et al. (2014), in addition to Kesler, Kent and O'hara (2011), who found no significant differences in the average performances in the task of the Digit Span, comparing post–chemotherapy women with healthy ones.
Another aspect that the present study identified referred to focused attention, assessed by the direct sequence Digit Span. The CQ group performed worse, agreeing with other studies such as Lyon et al. (2016), Dumas et al. (2013), Chen et al. (2014), Jansen, Cooper, Dodd and Miaskowski (2011), Zheng et al. (2014) and Piccirillo, et al. (2015). The research results of Dumas et al. (2013) and Piccirillo et al. (2015) showed decreased functional connectivity in the anterior dorsal attention network of these women, one month after chemotherapy. Such changes in connectivity corresponding to attention were also found in the study by Piccirillo, et al. (2015), pointing to a decrease in fractional anisotropy (FA) of white matter in frontal and temporal lobes. Park et al. (2015) also signaled attentional losses due to chemotherapy, obtaining improvements 6 months after the completion of treatment. However, no performance differences were found in the direct sequence Digit Span between the groups in the study by Chen et al. (2014).
Worse performance was also found in the Stroop test to assess reaction time in studies by Yao et al. (2016), Chen et al. (2014), Miao et al. (2016) and Koppelmans et al. (2012). In the last study, patients with chemotherapy showed significantly lower functional connectivity at the anterior cingulate cortex (ACC) and the most altered areas were found in the frontal lobes, temporal lobes and parietal lobes. The correlation analysis in this group also indicated a negative connection between the response times in the Stroop test and the strength of the functional connectivity of the ACC (Miao et al., 2016). Likewise, this was verified by Kam et al. (2015), where women after chemotherapy showed less dependent signal on the level of oxygen in the blood in several frontal regions, including the ACC, a critical region for the response inhibition.
In an analysis by Lyon (2016), groups of women who received chemotherapy at different times (4 weeks, 6 months, 1 year and 2 years) differed from groups of women with cancer without chemotherapy in terms of reaction time, cognitive flexibility and attention assessed by the Stroop Test. Over time, after 2 years of chemotherapy, there were high improvements (according to the baseline) only for reaction time, cognitive flexibility and executive functioning. For reaction time, moderate and minor improvements were observed for complex attention.
Kesler et al. (2011), it was observed that women undergoing chemotherapy demonstrated significantly reduced activation in the middle left of the dorsolateral prefrontal cortex and premotor cortex. They showed a significant reduction in left caudal lateral prefrontal cortex, associated with increased persistent errors and slower processing speed compared to the other two groups (women with breast cancer without chemotherapy and the control group). There were controversies, however, in the study by Jansen et al. (2011) and Kam et al. (2015), with no significant differences when it came to reaction time between the groups, noting that the latter found impaired differences in hemodynamic functioning, in relation to the care and inhibitory control system of women after chemotherapy.
The present study found no differences in terms of decision making. In contrast, in the study by Chen et al. (2014), the only one performed with the same profile of participants, the group of breast cancer patients undergoing chemotherapy had disadvantages in IGT in relation to the other two groups (without chemotherapy and control group), regarding the decision–making domain.
Longitudinal changes in the cognitive functioning of memory, attention and executive functions were detected in patients with breast cancer undergoing chemotherapy (Chen et al., 2014). According to Park et al. (2015), small improvements were seen 6 months after the completion of chemotherapy in cognitive domains of changing working memory. Zheng et al. (2014) found advances in immediate memory, delayed memory and attention within 6 months after chemotherapy. According to Dumas et al. (2013), in one year, the condition of functional connectivity corresponding to the attention that had been impaired partially returned to the baseline. Jansen et al. (2011) found improvements after 6 months in aspects of attention, however, did not identify longitudinal progress in working memory.
No studies were found that shared results on impulsivity related to chemotherapy, nor to the process of becoming ill with cancer. This may denote the need to encourage discussion on this aspect in the literature.
With regard to intervening variables, some studies have found considerable interferences in schooling, depression and fatigue in aspects of inhibitory control / cognitive flexibility (Lyon, 2016), in addition to disease severity, age and education (Kesler et al., 2011) . and fatigue (Askren et al., 2014) in general executive aspects. The present study ruled out the interference of age, education, anxiety and depression in post hoc analyzis.
Final considerations
This study verified the existence of cognitive impairments in women with breast cancer undergoing chemotherapy, with regard to important components of executive functioning such as attention, working memory, inhibitory control / cognitive flexibility and impulsivity assessed by direct and inverse sequence Digit Span tests, Stroop and BIS–11, respectively. In general, the data obtained corroborated with some research carried out under similar methods and in works with magnetic resonance neuroimaging.
No studies were found that track impulsiveness in this public. In addition, there is no consensus on aspects of decision–making, nor on their implications of cognitive variables in the therapeutic process of cancer, such as, for example, adherence to treatment. It should be noted that there are still gaps in the literature to explore the potential risk factors and predictors of cognitive changes related to chemotherapy. Psychological interventions to prevent and intervene in such cognitive disabilities should also be developed and tested. For this, it is also necessary to carry out studies of functional behavioral analysis that aim at more details about the contingencies involved during the process of diagnosis and treatment of cancer.
This research had limitations that must be considered, such as the transversality of data collection (not longitudinal), variation in the places of application of the tests, the low matching of the groups with respect to age, the non inclusion of qualitative data in self–reports, absence of ecological testing instruments and the lack of examination of other important covariables (such as fatigue and treatment adherence). On the other hand, the relevance of this study is reflected in the improvement of the identification of users who could benefit from neuropsychological services included in the complexity of Oncology. It is hoped, therefore, to foster discussion on assistance to women with breast cancer, aiming at an integrated chemotherapy and increasing this treatment with wide therapeutic possibilities, that is, a follow–up of care that considers, in addition to systemic side effects and disease survival, the quality of life in the daily activities of this user as well as in her social and family relationships.
References
Ahles, T. A., & Saykin, A. J. (2007). Candidate mechanisms for chemotherapy–induced cognitive changes. Nature Reviews Cancer, 7(3), 192–201. doi: 10.1038/nrc2073 [ Links ]
Ando–Tanabe, N., Iwamitsu, Y., Kuranami, H., Okazaki, S., Yasuda, H., Nakatani, Y., ... Miyaoka, H. (2014). Cognitive function in women with breast cancer receiving adjuvant chemotherapy and healthy controls. Breast Cancer Research and Treatment, 21(4), 453–462. doi: 10.1007/s12282–012–0405–7 [ Links ]
Askren, M. K., Jung, M., Berman, M. G., Zhang, H., Therrien, B., Peltier, S., ... Cimprich, B. (2014). Neuromarkers of fatigue and cognitive complaints following chemotherapy for breast cancer: a prospective fMRI investigation. Breast Cancer Research and Treatment, 147(2), 445–455. doi: 10.1007/s10549–014–3092–6 [ Links ]
Chen, X., Li, J., Zhu, C., Li, D., Zhang, J., & Wang, K. (2014). Cognitive function in breast cancer patients on chemotherapy: a longitudinal study. Zhonghua Yi Xue Za Zhi, 94(1), 27–30. doi: 10.1002/pon.1342 [ Links ]
Diamond, A. (2013). Executive functions. Annual Review Psychology, 64, 135–168. doi: 10.1146/annurev–psych–113011–143750 [ Links ]
Dumas, J. A., Makarewicz, J., Schaubhut, G. J., Devins, R., Albert, K., Dittus, K., & Newhouse, P. A. (2013). Chemotherapy altered brain functional connectivity in women with breast cancer: A pilot study. Brain Imaging and Behavior, 7(4), 1–16. doi: 10.1007/s11682–013–9244–1 [ Links ]
Ganz, P. A., Kwan, L., Castellon, S. A., Oppenheim, A., Bower, J. E., Silverman, D. H., ... Belin, T. R. (2013). Cognitive complaints after breast cancer treatments: Examining the relationship with Neuropsychological Test Performance. Journal of the National Cancer Institute, 105(11), 791–801. doi: 10.1093/jnci/djt073 [ Links ]
Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). (2012). Ensino em atenção oncológica no Brasil: carências e oportunidades. Ministério da Saúde. Rio de Janeiro: Autor. Recuperado de http://www1.inca.gov.br/inca/Arquivos/ensino_em_atencao_oncologica_no_brasill.pdf [ Links ]
Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). (2016). Quimioterapia. Rio de Janeiro: Autor. Recuperado de http://www.inca.gov.br/conteudo_view.asp?ID=101 [ Links ]
Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). (2019). Estimativa 2020: incidência de câncer no Brasil. Rio de Janeiro: Autor. [ Links ]
Jansen, C. E., Cooper, B. A., Dodd, M. J., & Miaskowski, C. A. (2011). A prospective longitudinal study of chemotherapy–induced cognitive changes in breast cancer patients. Support Care Cancer, 19(10), 1647–1656. doi: 10.1007/s00520–010–0997–4 [ Links ]
Jemal, A., Vineis, P., Bray, F., Torre, L., & Forman, D. (Orgs.) (2014). The Cancer Atlas (2nd ed.) Atlanta, GA: American Cancer Society. Recuperado de www.cancer.org/canceratlas [ Links ]
Kam, J. W., Boyd, Â L. A., Hsu, C. L., Liu–Ambrose, T., Handy, T. C., Lim, H. J., ... Campbell, K. L. (2015). Altered neural activation during prepotent response inhibition in breast cancer survivors treated with chemotherapy: An fMRI Study. Brain Imaging and Behavior, 10(3). 1–9. doi: 10.1007/s11682–015–9464–7 [ Links ]
Kesler, S. R., Kent, J. S., & O'hara, R. (2011). Prefrontal cortex and executive function impairments in primary breast cancer. Archives of Neurology, 68(11), 1447–1453. doi: 10.1001/archneurol.2011.245 [ Links ]
Koppelmans, V., Breteler, M. M., Boogerd, W., Seynaeve, C., Gundy, C., & Schagen, S. B. (2012). Neuropsychological performance in survivors of breast cancer more than 20 years after adjuvant chemotherapy. Journal of Clinical Oncology, 30(10), 1080–1086. doi: 10.1200/JCO.2011.37.0189 [ Links ]
Lyon, D. E., Cohen, R., Chen, H., Kelly, D. L., Starkweathe, U. M., Ahn, H. C., & Jackson–Cook, C. K. (2016). The relationship of cognitive performance to concurrent symptoms, cancer and cancer treatment related variables in women with early stage breast cancer: A 2 year longitudinal Study. Journal of Cancer Research of Clinical Oncology, 142(7), 1–14. doi: 10.1007/s00432–016–2163–y [ Links ]
Malloy–Diniz, L. F., Leite, W. B., Moraes, P. H. P., Correa, H., Bechara, A., & Fuentes, D. (2008). Versão em português do Brasil da Iowa Gambling Task: adaptação transcultural e validade discriminante. Revista Brasileira de Psiquiatria, 30(2), 144–148. doi: 10.1590/S1516–44462008005000009 [ Links ]
Malloy–Diniz, L. F., Mattos, P., Leite, W. B., Abreu, N., Coutinho, G., Paula, J. J., ... Fuentes, D. (2010). Tradução e adaptação cultural da Barratt Impulsiveness Scale (BIS–11) para aplicação em adultos brasileiros. Jornal Brasileiro de Psiquiatria, 59(2), 99–105. doi: 10.1590/S0047–20852010000200004 [ Links ]
Marcolino, J. A. M, Mathias, L. A. S. T, Piccinini, L., Guaratini, A. A., Suzuki, F. M., & Alli, L. A. C. (2007). Escala Hospitalar de Ansiedade e Depressão: estudo da validade de critério e da confiabilidade com pacientes no pré–operatório. Revista Brasileira de Anestesiologia, 57(1), 52–62. doi: 10.1590/S0034–70942007000100006 [ Links ]
McDonald, B. C., Conroy, S. K., Smith, D. J., Oeste, J. D., & Saykin, A. J. (2013). Frontal gray matter reduction after breast cancer chemotherapy and association with executive symptoms: A replication and extension study. Brain, Behavior, and Immunity, 30, 117–125. doi: 10.1016/j.bbi.2012.05.007 [ Links ]
Miao, H., Li, J., Hu, S., He, X., Partridge, S. C., Ren, J., ... Qiu, B. (2016). Long–term cognitive impairment of breast cancer patients after chemotherapy: A functional MRI study. European Journal of Radiology, 85(6), 1053–1057. doi: 10.1016/j.ejrad.2016.03.011 [ Links ]
Miller, E. K.; Freedman, D. J.; & Waliis, J. D. (2002). The prefrontal cortex: Categories, concepts and cognition. Philosophical Transations of the Royal Society London B: Biological Sciences, 357, 1123–1136, 2002. doi: 10,1098/rstb.2002.1099 [ Links ]
Nauta, W. J. H. (1971). The problem of the frontal lobe: A reinterpretation. Journal of Psychiatric Research, 8(3–4), 167–187. doi: 10.1016/0022–3956(71)90017–3 [ Links ]
Park, J. H., Bae, S. H., Jung, Y. S., & Jung, Y. M. (2015). Prevalence and characteristics of chemotherapy–related cognitive impairment in patients with breast cancer. Journal of Korean Academy of Nursing, 45(1), 118–128. doi: 10.4040/jkan.2015.45.1.118 [ Links ]
Piccirillo, J. F., Hardin, F. M., Nicklaus, J., Kallogjeri, D., Wilson, M., Ma, C. X., ... Schlaggar, B. L. (2015). Cognitive impairment after chemotherapy related to atypical network architecture for executive control. Oncology, 88(6), 360–368. doi: 10.1159/000370117 [ Links ]
Salvo, L., & Castro, A. (2013). Confiabilidad y validez de la escala de impulsividad de Barratt (BIS–11) en adolescentes. Revista Chilena de Neuro–Psiquiatria, 51(4), 245–254. doi: 10.4067/S0717–92272013000400003 [ Links ]
Snaith, R. P. (2003). The Hospital Anxiety And Depression Scale. Health and Quality of Life Outcomes, 1, 1–29. doi: 10.1186/1477–7525–1–29 [ Links ]
Strauss, E. A. (2006). Compendium of neuropsychological tests: Administration, norms and commentary (3rd. ed.). New York: Oxford University Press. [ Links ]
Yao, C., Rich, J. B., Tannock, I. F., Seruga, B., Tirona, K., & Bernstein, L. J. (2016). Pretreatment differences in iintraindividual variability in reaction time between women diagnosed with breast cancer and healthy controls. Journal of the International Neuropsychological Society, 22, 530–539. doi: 10.1017/S1355617716000126 [ Links ]
Zheng, Y., Jianfeng, L., Pingping, B., Hui, C., Zhen, H., Ding, , ... Qi, D. (2014). Long–term cognitive function change among breast cancer Survivors. Breast Cancer Research and Treatment, 146(3), 599–609. doi: 10.1007/s10549–014–3044–1 [ Links ]
Recepted in 14.jun.18
Revised in 22.mar.20
Accepted in 17.apr.20
Camila Vasconcelos Carnaúba Lima, Mestre em Psicologia pela Universidade Federal de Alagoas – UFAL, é Professora na Universidade Tiradentes – UNIT. Endereço para correspondência: Rua Artur Vital da Silva, 573, Ed. San Martin, apt. 304, Gruta de Lourdes, Maceió â AL. 57.052–790. Email: camilacarnauba@gmail.com
Rosa Maria Martins de Almeida, Doutora em Fisiologia pela Universidade Federal do Rio Grande do Sul – UFRGS, Pós–Doutora em Neuropsicofarmacologia pela Tufts University, Boston, USA, é Professora Associada na Universidade Federal do Rio Grande do Sul – UFRGS. Email: rosa_almeida@yahoo.com
Raner Miguel Ferreira Póvoa, Doutor em Ciências pela Universidade Federal do Espírito Santo – UFES, é Professor da Universidade Federal de Alagoas – UFAL. Email: povoaraner05@gmail.com
1 To guarantee participant's confidentiality, such institurions will not be disclodes.














