Serviços Personalizados
artigo
Indicadores
Compartilhar
Revista de Etologia
versão impressa ISSN 1517-2805versão On-line ISSN 2175-3636
Rev. etol. v.6 n.2 São Paulo dez. 2004
ARTIGOS
On the panic reactions of ants to a crushed conspecific: a contribution to a psychoethology of fear*
Sobre as respostas de pânico de formigas diante de uma coespecífica esmagada: contribuição para uma psicoetologia do medo
Walter Hugo de Andrade Cunha
I University of São Paulo - Brazil
ABSTRACT
By crushing an ant on a trail of Paratrechina (Nylanderia) fulva Mayr; ants one causes marked changes in the behavior of the remaining ants when they come near the dead conspecific which may be called panic reactions (detour, going back, waving march, jolt and disorientation) and which represent disturbances or a disorganization of the prevailing behavior in the trail (Cunha, 1967). In the first part of the present paper, an attempt is made to show how the form and specific circumstances of each of such reactions may be the result of previous experience of the ants with the situation to be changed. In the second part, using data from a few species of four ant subfamilies (Myrmicinae, Formicinae, Ponerinae and Dorylinae) on the intensity of reaction to a crushed conspecific on the trail, it is shown that intensity of disturbance appears to be correlated not with taxonomic position but with the degree of mutual attraction or interaction at an individual level present in each species. Fear is reinterpreted as a motivacional state characterized by an intensified process of sensory discrimination and by the simultaneous presence of two tendencies: search for the familiar and flight from the unfamiliar.
Index terms: Alarm behavior, Fear, Ants, Paratrechina (Nylanderia) fulva.
RESUMO
Esmagando-se uma ou mais formigas sobre a trilha da formiga Paratrechina (Nylanderia) fulva Mayr, provocam-se mudanças marcadas no comportamento das outras quando chegam perto da coespecífica morta. Estas "reações de pânico" (desvio, retorno, marcha ondulante, sacolejo e desorientação) representam uma perturbação ou uma desorganização do comportamento que prevalecia na trilha antes da mudança (Cunha, 1967). Na primeira parte do presente trabalho, tentou-se mostrar como a forma e as circunstâncias específicas de cada uma dessas reações são um reflexo da experiência prévia das formigas individuais com a situação posteriormente alterada. Na segunda parte, utilizando-se dados de algumas espécies de formigas de quatro sub-famílias (Myrmicinae, Formicinae, Ponerinae e Dorylinae) sobre a intensidade da reação a coespecíficas esmagadas na trilha, mostrou-se como esta intensidade parece correlacionada, não com a posição taxonômica das espécies consideradas, mas com o grau em que os indivíduos dessas espécies são capazes de atração mútua e de interação num nível propriamente individual. O medo é reinterpretado como um estado motivacional caracterizado por um processo de intensa discriminação sensorial e por duas tendências simultâneas: uma de busca do familiar e outra de fuga do infamiliar.
Descritores: Comportamento de alarme, Medo, Formigas, Paratrechina (Nylanderia) fulva.
By crushing (Cunha, 1967) one or more ants on a trail of Paratrechina (Nylanderia) fulva Mayr ants, one usually causes marked changes in the behavior of the remaining ants, when they come near the killed companions. These behavior changes (see some categories below) will here be collectively called, by convenience, "panic reactions".
By means of several experiments, we hope to have demonstrated that these reactions are a result of a discrepancy between the situation the ants were used to and this same situation as they find it after some experimental alteration. The fact that a crushed companion on a trail is usually the best means to elicit panic reactions in ants is probably due to the exceptionally good opportunities that an ant finds to get familiar with or conditioned to these objects in its normal life.
A very similar interpretation - a conflict between behavior tendencies aroused by a perceptual conflict involving familiarity-infamiliarity with a given situation - had already been advanced by Hebb (1946) to explain what he called fear in chimpanzees: the fact that these animals reacted with shrill vocalizations, pill erection, etc., to some subjects which were, by the first time, shown to them. Specially efficient were, to this effect, objects representing primates with some gross deformation, as, for instance, a chimp's head detached from the body with moveable jaws, a facial chimp mask, etc. These phenomena, Hebb argued, were similar to the human horror of a mutilated person, and were to be classed together with other reactions in a class of "fear of the strange"phenomena. Hebb also developed a neurological interpretation for these phenomena which will not concern us here.
As Hebb already pointed out, the fear reactions considered show aspects which allow us to classify them, as to the source of their determination, both as acquired and as innate phenomena. In fact, they do not seem to occur in the absence of previous experience with the situation which will later on be altered, and so they would classify as acquired. On the other hand, they do not result from training or gradual reinforcement due to the operation of known laws of learning. Instead, they manifest themselves in their complete form from their very first occurrence, and so would also classify as innate.
In this paper, we will present data related to the above mentioned assumptions about the dual origin of the factors affecting ants panic reactions.
Possible relationship between panic reactions and individual experience
We were unable to provide our ants with individual marks, in the field, so we had to use indirect evidence to show that panic reactions are determined by factors originating in the past experience of the ants. For this purpose, we chose to present and to discuss here the intercorrelations obtained along the more systematically recorded observational categories in our studies (Cunha, 1967). These are the categories without apparent disturbance (which is taken to represent base line behavior) and the behavior categories called passage, pause, detour, going back, disorientation, waving march and jolt.
Prior to examining the above mentioned relationship, let us point out briefly what we assume might be the psychological determinants of each category. In the first place, we must say that the experimental alteration (for instance, a crushed ant) generally functions as an aversive obstacle in the situation. And we must say also, in this connection, a passage would seem to indicate a lesser deterrent character in an "obstacle" than a pause, and this latter still less than a detour, and this, in turn, still less than a going back.
In second place, let us consider each behavior category separately, taking into account its form and circumstances. A passage (march along the trail) is base line behavior. It may, however, occur after one interferes mildly in a trail (for instance, projecting a light spot on it, or putting an object outside of it). A pause (temporary suspension of locomotion) is considered to be an indication of a conflict of the ant between going and not going on its previous path. In fact, we can, for instance, see frequent pausing of ants in front of food which we use as an obstacle in an intermediate point of the trail where it seems to be both an attractive and an aversive interferent object.
A detour seems to be a compromise between escape and going on. Because it involves marching outside the trail, it seems to imply good orientation capacity and little dependence on the trail for purposes of orientation. Perhaps it indicates an ant that was among the establishers of the trail, when light or wind direction, or gravitational cues, could or should be attended to. To the contrary, a disorientation - the slow and continually reorientated march of an ant which goes out of the trail - seems to imply just the opposite of a detour. Perhaps it denotes an ant which has come to walk in the trail only after it had been consolidated, or only for a limited number of times.
A going back, in our experiments, seems to have two main components. The first one is escape, characterized by speeded locomotion away from the unfamiliar elements in the situation. The second one is the fact that the ant persists in its locomotion away from the altered enviroment till it finds a region - which tends to be one to which the ant is most familiar - where it immobilizes or gives itself up to comfort movements. These two components suggest that the going back results from a perceptual conflict involving familiar and unfamiliar elements, and which is so strong that it arouses in the ant a new, changed motivational state which we will call fear. Fear, then - and in this we extend the hebbian conception - is seen as a motivational state composed of two simultaneous tendencies: one of escaping the unfamiliar and/or the aversive elements in a situation, and another, which the layman identifies as "search for refuge", and which, perhaps, is only, in the final analysis, but an expression of a tendency to let behavior to be so more guided by a given element in the situation how more this element is familiar and/or gratifying or associated with gratification in the past. As such, this state also implies, necessarily, an intensified sensorial discrimination, which, in our ant, we believe is expressed by that kind of oscillation seen in the waving march which generally accompanies flight from the experimental alteration. Now, what kind of confict situation, or what elements in such a situation, would favor the motivational change represented by intense fear? - we think that these are the opposite of elements which favour a detour, e.g., strong familiarity with the trail, and great dependence on it for purposes of orientation.
A waving march seems, besides what was said above, to be a composite category formed by the impinging, upon locomotion, of frontal oscillation in the ant's body from side to side. As this pattern seems instrumental for localizing some aspects of the enviroment - e.g., the lost trail or an experimenter's finger put above one side of a resting ant - we suppose that its occurrence together with locomotion expresses a state of intensified discrimination. We must admit that it was the waving element in a flight that led us initially to think of a disturbed ant as being in a state of what humans call panic. Perhaps this impression may not be wrong at all: if a flight, speeded as it may be, is an organized instead of a vacillating affair, it may not contain fear at all, as Hebb (1946, 1949) had already pointed out. In this way fear may again be said to have in its central chore a motivational state of escape from the unfamiliar and search for the familiar.
A jolt is a sudden, jerky backwards movement of the ant's body, on stiff legs. Because of its suddenness and its short duration, it reminds us of the startle reflex of mammals, which is considered to be a pattern of unconditioned responses to a sudden alteration in a situation (see, for instance, Keller & Schoenfeld, 1950). In fact, many unexpected events or objects which appear suddenly in front of an ant may cause it to jolt. Also one can find a jolt more easily by disturbing an old trail than a recent one. Those facts suggest that a jolt may also imply a strong conditioning to or absorption in the situation which is being altered. It might be that an ant susceptible to jolt was so stereotyped in its journeys along the trail, or so old in it and dependent on it for orientation purposes that it would be, so to say, unprepared to face something new on it.
After these interpretive comments, let us choose one relationship, amog others obtained in a previous work (Cunha, 1967), to examine in this paper. This will be the correlation (Pearson r) between the above mentioned categories, the correlated variable being the percentage of ants in each category. For this purpose, we pooled the records of 14 observations in which dead conspecifics were employed - intact or crushed in the whole or in some part, in the trail, or outside of it - as an experimental alteration. One should note that, in this way, what the coefficients tell us is whether the factors, whatever they be, that tend to determine the occurrence of a given category are or are not the same that tend to determine the occurrence of any other of the considered categories. Table 1 shows the results.
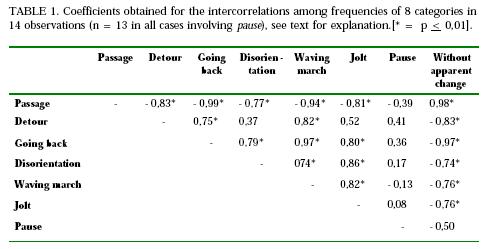
Let us first point that the correlation between passage and without apparent change is positive and pratically perfect. This result seems to justify assuming that a passage is normal, undisturbed behavior. On the second hand, let us note that the correlation coefficients between either passage or without apparent change and all the remaining categories are negative, and - except for the case of pause - statistically signifficant. This shows that the factors that determine passage and without apparent change tend to be the same that work against the determinations of the remaining ones, except pause. In other words, these results show us that detour, going back, waving march, jolt and disorientation are disturbances or a disorganization of the behavior that was occuring in a trail before we interfered with it, whereas the nature of the disturbance of a pause is by no means clear. This is in line with our interpretation of these categories.
The correlation coefficients between going back, detour and pause, on one hand, and without apparent change and passage on the other show decreasing values, in that order. This, we remember, is just what was expected. Notice also, that waving march has a high negative correlation with passage and without apparent change, whereas is is almost perfectly and positively correlated with going back. This seems to give us some justification for considering waving march, together with going back, a severe behavior disturbance in the context of our observations.
Pause is the only category in our list of categories of behavior changes which does not correlate significantly with any other category. This suggests that the factors which determine these categories do not determine a pause, which appears, thus, as a sui generis category. Figure 1 may give us a hint of why this is so.
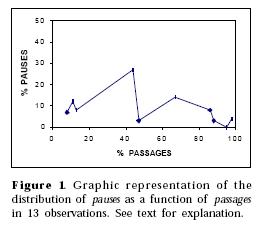
In Figure 1, we see how the pauses are distributed as a function of passages in 13 observations in our study. If we take the amount of passages in each observation as an inverse index of the degree to which the experimental alteration in that observation functions as an aversive obstacle to the ants, we can see that there is a tendency for more ants to be pausing when this value is moderate than when it is either strong, or, conversely, weak - when, in terms of our interpretation, going back and passage should, respectively, become more likely to occur. The same type of curve was found to represent the distribution of pauses as a function of any other categories of behavior change. This seems in line with our interpretation, which implies a linear representation of all these categories, except pause, on the degree of aversiveness of the experimental alteration.
Considering this common property, it is natural, also, to expect covariation of the categories going back, waving march, detour, jolt and disorientation. The fact that most of their intercorrelations, shown in Table 1, turn out to be positive and statistically significant will, then, be a normal result, in terms of our interpretation, and will not require a special commentary. In what follows, we will, then, restrain our comments to the relationships obtained between the category detour, on one hand, and the categories disorientation and jolt, on the other. These relationshps are expressed, in Table 1, by small, non- significant positive correlations. This is so, we think, because the factors of trail dependence and knowledge of alternative routes - which, according to our interpretation, are very different in the detour and the jolt categories - work against the positive correlation contributed by the common factor of aversive determination. Thus, even in these cases, we seem to find out additional support for our interpretation.
We think we have, then, in all the above considerations, a reasonable support for the contention that one can find, in the individual experiences of each ant, some of the factors that determine the particular way in which it will react to an adultered conspecific in connection with a trail. We turn now to a consideration of the other possibility present in Hebb's thinking.
Possible determinants of panic reaction by factors introduced by the species of the ants concerned:
Was it mere coincidence that adultered conspecifics had been found to be the most adequate objects to elicit panic reactions in ants, chimpanzees, and - so it seems - in human beings too? Or would this fact be somehow linked to the social nature of the three kinds of animals considered? (To these we should, certainly, include fishes (Minnows, for instance, according to a finding of Von Frisch cited in Tinbergen, 1953, who dash to cover to the scent of a skin substance of an injured conspecific) and, as we could observe in some cases, domestic cattle, woodpeckers, house sparrows and domestic fowls: all, one should notice, social species.)
The data we have concerning this topic resulted from a brief attempt we made to appreciate the generality of the panic reactions in other species of ants besides P. fulva. For this purpose, we observed the reactions of the workers of several species of ants of four unequally represented sub-families to the crushed bodies of conspecifics on a trail, and, sometimes, in other circumstances as well (Cunha, 1967). In Table 2 we present a classification of the species considered according to the degree - mild, moderate, or high - of the panic reactions that were observed. We also ordered the species in each class, placing the less disturbed at the top, and the more disturbed at the bottom of each column. The basis for the classification was sheer impression of the general character of the reactions. We found that only some species among the Myrmicinae and the Formicinae exibited reactions approaching the catastrophic character of those shown by P. fulva. From the other side, the species with the mildest reactions were T. marginata, which is a Ponerinae, and the two species of Labidus, which are Dorylinae.
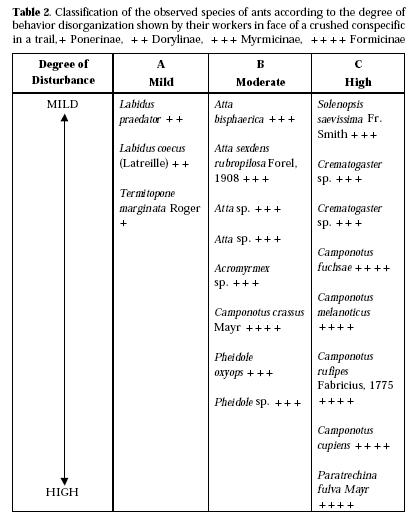
Are these differences occasional, or, instead, do they correspont to factors introduced by the taxonomic position of these species? Admitting with some authors (see, for instarnce, Forel, 1921; Wheeler, 1910) that ant evolution was greatly presided by social adaptation; considering the apparent importance of familiarity of the normal companion's body for the phenomena being dealed with here; and considering also plausible that social evolution could affect, by determining the social organization of each group, the manner in which the ants might interact and get familiar with each other, we consider the question above sufficiently meaningful to be followed even with such scarce sample of species as we have here. If we do, and if we compare such a picture with that given us on ant social and structural evolution by Wheeler (1910) and Forel (1921), we will find a certain amount of correspondence in them. Thus, the four subfamilies represented in our study will be ordered in this way, according to these authors, from more primive to more advanced both in structural and social characters: Ponerinae, Dorylinae, Myrmicinae and Formicinae. Only the first two are represented in column C, Table 2. Besides, the fact that our Formicinae, as a group, have shown more panic than the Myrmicinae, and these, in turn, more than the Ponerinae and the Dorylinae would add some weight to the hypothesis of a correspondence. However, this is contradicted by the fact that T. marginata - a ponerine - has shown more severe disturbance than the two species of Labidus, which are Dorylinae. Also, the picture concerning the Myrmicinae and the Formicinae is not entirely what one should have predicted.
We find still more difficult to see a clear correspondence between the classification presented in Table 2 and the phylogenetic diagram implied in the more modern version of ant evolution developed in 1954 by Brown (cited by Wilson, 1971). This version assumes that ants derived independently twice from the ancestor tiphioid wasp, and these different origins are now represented by the myrmecioid and poneroid complexes of ant sub-families.
To the myrmecioid complex belongs the Formicinae ants in our table, and to the poneroid the remaining ones. Now, nothing in Table 2 reminds us of such a clear separation. Perhaps this would be so because true sociality in ants may have arisen only once, and this could have occurred before the aforementioned branching. But the question of how many times and when eusociality originated independently in ants is considered very conjectural by Wilson (1971).
Be it as it may, we wish to present an alternative to the above attempt to see a direct correspondence between taxonomic position, based either in social or structural atributes, and severity of panic reactions in face of an adultered conspecific. It seems to us - on the basis of many facts presented by Morley (1954) - that it would be possible for a given species to be socially more advanced that other (for example, in having a more complex social organization) without being superior to that other in such matters, as for instance, degree of mutual attraction or intimacy in the intereactions of its members. In this connection, it is appropriate to poit out another fact revealed by previous studies (Cunha, 1967): the fact that the crushing of P. fulva's head is more effective than that of its thorax, and this, in its turn, more than that of its gaster to elicit panic reactions in this species of ant. According to our data, these differences seem to be due, not to differences of amount of pherormone released by crushing per se, but more probably, to the differences in the degree that these anatomical regions would be familiar to the ants as a result of previous social contacts, specially during trophallaxis (in the sense of exchange of regurgitated crop liquids). Trophallaxis, because it implies intimate and prolonged bodily contacts, seems to offer a specially good opportunity, during ontogeny, for an ant to become familiar with the characteristics of a companion's body, and, thus, to become specially apt to react with panic to such a body when adultered. All this suggests that trophallaxis might be the key we were in search of, for explaining the differences presented in Table 2. In fact, in this very behavior the Ponerinae are, admitedly, superior to the Dorylinae, and clearly surpassed by the Mymicinae and, above all, by the Formicinae. More than that, we think we can even explain most of the intra-classes differences in Table 2 on the basis of what is known of relative importance of liquid regurgitation in the species concerned. So, Crematogaster was, for instance, among the Myrmicinae in Table 2, the most severely disturbed group. Being an aphid tending genus, it is also, among them, the one which indulges more in reciprocal regurgitation. S. saevissima was the next more disturbed Myrmicinae. It also tends aphids, although not so exclusively as Crematogaster. Next came the species of Pheidole, and, finally, species of Atta and Acromyrmex. Now, Pheidole ants are generally either insectivorous or gravivorous, whereas Atta and Acromyrmex, are fungus growers, and all these habits, according to Morley (1954, p. 110), explain why the mechanism of regurgitation has never become much developed in these groups.
Let us consider, now, the Formicinae ants in Table 2. P. fulva is the one which exhibits the more dramatic panic reactions among this group, as well as among the species of the whole table. It is also characterized by hypertrophy in the regurgitation functions, being considered, by its specialization in crop distension - a trace connected with trophallaxis - as a "temporary replete ant" (See, in this connection, Wheeler, 1910, Ch. XX; Forel, 1922, Vol. III, p. 78; Michener & Michener, 1953, ch. 18; Morley, 1954, p. 110). The remaining Formicinae in our list, all of the genus Camponotus, are herding ants and, for that matter, make heavy use of trophallaxis. The fact that they are - except for one species - in column C is understandable, according to our hypothesis. But how to explain the exception represented by C. crassus? - The reason for this, we think, lies in the fact that C. crassus, differently from all other ants in Table 2, save, perhaps, T. marginata, is endowed with relatively good vision (it is a diurnal ant). This makes it much less dependent on the trail than its almost blind colleagues - a factor that, as we saw when we considered the category detour, is very important for lessening the panic induced by a crushed conspecific on a trail.
In summary, we think we may have, in our results and considerations, in spite of all their limitations, at least a promising hypothesis from which to start, in our attempt to discover possible links between ant's panic reactions and factors inherent in the social organization of their species. We hope that this hypothesis, and the small amount of facts connected to it, may constitute, together with the precedent findings and considerations concerning experimental effects, a contribution to what we could perhaps dare to call a psychoethology of fear.
References
Cunha, W. H. de A. (1967). Explorações no mundo psicológico das formigas. 1 - Uma investigação de algumas modificações de comportamento de formigas relativamente ao verificado numa situação inalterada anterior, com especial referência a Paratrechina (Nylanderia) fulva Mayr (Hymenoptera-Formicidae) (2 Vols.). Tese de Doutorado, Instituto de Psicologia, Universidade de São Paulo, São Paulo. [ Links ]
Forel, A. (1921-1923). Le monde social des fourmis du globe comparé à celui de l'homme (5 Vols.). Genèbre: Librairie Kundig. [ Links ]
Guilford, J. P. (1965). Fundamental statistics in psychology and education (4th ed.) New York: McCraw-Hill. [ Links ]
Hebb, D. O. (1946). On the nature of fear. Psychological Review, 53, 259-276. [ Links ]
Hebb, D. O. (1949). The organization of behavior: A neuropsychological theory. New York: Wiley. [ Links ]
Keller, F. S., & Schoenfeld, W. N. (1950). Principles of psychology. New York: Appleton-Century-Crofts. [ Links ]
Michener, C. D., & Michener, M. H. (1951). American social insects. New York: Van Nostrand. [ Links ]
Morley, D. W. (1954). The evolution of an insect society. London: George Allen and Unwin. [ Links ]
Tinbegen, N. (1953). Social behavior in animals, with special reference to vertebrates. London: Methuen. [ Links ]
Tolman, E. C. (1932). Puposive behavior in animals and men. Berkeley: University of California Press. [ Links ]
Wheeler, W. M. (1910). Ants: Their structure, development, and behavior. New York: University of Columbia Press (4th printing, 1965). [ Links ]
Wilson, E. O. (1971). The insect societies. Cambridge, MA: The Beldnap Press of Harv. Univer. Press. [ Links ]
 Correspondence
Correspondence
Walter Hugo de Andrade Cunha
R Martiniano de Carvalho, 676 - Ap 41, Paraiso
São Paulo, Brazil
E-mail: whac@uol.com.br
Received September ??, ????
Revision received November ??, ????
Accepted March, ????
* This work is part of a paper read at the symposium "Ontogeny and Phylogeny of Behavior", during the I Latinamerican Congress of Psychobiology, held in São Paulo, Brazil, in 1973. I thank Mr. Gordon P. Bodely for the English translation of this article and Dr. César Ades for the translation of the French abstract (not included).














