INTRODUCTION
The aging population presents significant challenges to public health, particularly concerning the increasing prevalence of chronic conditions such as cognitive decline[1], neurodegenerative diseases, and metabolic disorders[2]. As life expectancy continues to rise, so does the burden of age-related conditions, including Alzheimer’s disease, Parkinson’s disease, diabetes, cardiovascular diseases, and hypertension[3]. These conditions not only compromise the quality of life for elderly individuals but also pose economic and healthcare system burdens. Given the growing need for effective and accessible interventions, there is considerable interest in non-pharmacological strategies that may mitigate these health concerns and support cognitive and metabolic health in aging populations[4].
Among these interventions, probiotics have emerged as a promising area of research. Probiotics are live microorganisms that, when administered in adequate amounts, provide health benefits by modulating the gut microbiota[5]. Increasing evidence suggests that probiotics play a role in cognitive and metabolic health, potentially influencing neuroprotection, inflammation, insulin sensitivity, and lipid metabolism. Studies indicate that probiotic supplementation may help maintain cognitive function[6], reduce neuroinflammation, and support gut-brain axis communication, which could be particularly beneficial in preventing or slowing neurodegenerative diseases[7]. Additionally, probiotics have been associated with metabolic improvements, including glycemic control, lipid profile regulation, and blood pressure management[8]. Despite these potential benefits, the extent and consistency of these effects remain uncertain due to variations in study design, probiotic strains, dosage, and duration of interventions[9].
To address these gaps, this study aims to systematically evaluate the cognitive and metabolic effects of probiotic supplementation in elderly individuals through an umbrella review with meta-analysis and evidence mapping. By synthesizing existing evidence from systematic reviews and meta-analyses, this research seeks to provide a comprehensive understanding of the potential benefits and limitations of probiotic use in aging populations. Furthermore, this study will assess the methodological quality of existing reviews, explore sources of heterogeneity, and identify research gaps that need to be addressed in future studies. By consolidating findings from multiple sources, this umbrella review aims to contribute to evidence-based recommendations for the use of probiotics in the promotion of cognitive and metabolic health among older adults. Therefore, this umbrella review with meta-analysis aims to synthesize existing evidence on the cognitive and metabolic effects of probiotics in older adults, while assessing the quality of the evidence and identifying gaps to guide future research and clinical recommendations.
METHODS
A Meta-umbrella review was conducted in accordance with the JBI methodology and the Reporting Guideline for Overviews of Interventions in Health (PRIOR) [10]. To ensure comprehensive reporting, the review followed the Preferred Reporting Items for Systematic Review and Meta Analysis Protocols (PRISMA-P) guidelines [11] and was prospectively registered in Open Science Framework DOI 10.17605/OSF.IO/ENJ8U.
A systematic search was performed in Web of Science, PubMed, Scopus, EMBASE, Cochrane, Epistemonikos, and CINAHL. Based on the established research question, we developed a preliminary search strategy for Medline/Pubmed using the ECUs (Extraction, Conversion, Combination and Use) method steps. To ensure the feasibility of the method, we followed the guidelines of the Peer Review of Electronic Search Strategies (PRESS) checklist [12].
What is the effectiveness of probiotics in improving cognitive and metabolic outcomes in elderly individuals with Diabetes Mellitus, cardiovascular diseases, Alzheimer’s, Parkinson’s and depression?
The PICO(S) framework used in this study aimed to assess the effects of probiotic supplementation in elderly individuals with chronic diseases. The target population included older adults with Diabetes Mellitus, cardiovascular diseases, Alzheimer’s, Parkinson’s, and depression, conditions that significantly impact public health and are often associated with cognitive and metabolic decline. The intervention analyzed was probiotic supplementation, including different strains and formulations, investigating its influence on gut microbiota and potential connections to cognitive and metabolic functions.
As a comparison, the study considered standard treatments, including usual care and conventional therapies for these conditions, to evaluate whether probiotics provide additional benefits beyond traditional approaches. The outcomes examined included cognitive effects, such as improvements in mental function and the prevention of neurodegenerative diseases, and metabolic effects, such as glycemic control, lipid profile regulation, and blood pressure management. To ensure methodological rigor, the study exclusively included systematic reviews and meta-analyses, offering a consolidated perspective on the available literature.
The search strategy was designed to capture a wide range of relevant studies by combining specific terms for each PICO(S) component. The population was identified using terms such as “elderly,” “older adults,” “aged,” and “seniors”, linked to conditions like diabetes, cardiovascular diseases, Alzheimer’s, Parkinson’s, and depression. The intervention was searched using descriptors like “probiotics,” “probiotic supplements,” “probiotic therapy,” and “probiotic treatment”. For the comparison, terms such as “standard care,” “usual care,” and “conventional treatment” were included. The outcomes were mapped using terms such as “cognitive effects,” “cognitive function,” “metabolic effects,” and “metabolic health”, ensuring the inclusion of studies evaluating these impacts. The study types were identified with descriptors such as “review,” “systematic review,” “meta-analysis,” “overview,” and “scoping review”.
To ensure a comprehensive approach, the search strategy should include terms related to the various contexts of the health conditions involved. The final search was:
(“elderly” OR “older adults” OR “aged” OR “seniors”) AND (“diabetes mellitus” OR diabetes OR “cardiovascular diseases” OR “cardiovascular disease” OR “Alzheimer’s disease” OR “Alzheimer’s” OR “Parkinson’s disease” OR “Parkinson” OR “depression” OR “depressed patients”) AND (“probiotics” OR “probiotic supplements” OR “probiotic therapy” OR “probiotic treatment”) AND (review OR “systematic review” OR “systematic literature review” OR “meta-analysis” OR “meta analysis” OR overview OR “rapid review” OR “integrative review” OR “scoping review”)
To develop the search strategy, four controlled vocabularies were used: Medical Subject Headings (MeSH), EMTREE from Embase, CINAHL Subject Headings and Health Science Descriptors (DeCS). These controlled terms were combined with free terms in natural language together with the Boolean operators AND and OR to increase the sensitivity of the search and obtain a more comprehensive spectrum of relevant results [13].
The inclusion criteria ensured the selection of high-quality studies that aligned with the research objectives. Only systematic reviews with meta-analyses evaluating the cognitive and metabolic effects of probiotic supplementation in older adults were included. Studies had to assess relevant health outcomes, such as cognitive function, glycemic control, lipid profile, blood pressure, and inflammatory markers. No restrictions were applied regarding language or year of publication, ensuring a comprehensive evaluation of the available evidence. Additionally, included studies had to follow a clear methodology, including a well-defined search strategy, explicit inclusion criteria, and a quantitative synthesis of results. Only reviews that included human participants and provided a rigorous evaluation of the data were selected.
The exclusion criteria were applied to ensure that only the most relevant and methodologically rigorous studies were included in the final analysis. Studies were excluded if they did not follow a systematic review with meta-analysis design, ensuring that only comprehensive syntheses of evidence were considered. Research that focused on populations outside the scope, such as studies that did not specifically assess older adults, was also removed. Additionally, studies evaluating interventions other than probiotics, including prebiotics, symbiotics, or general dietary modifications, were excluded. To maintain the focus on cognitive and metabolic effects, studies that assessed unrelated outcomes were not considered. Low methodological quality was another exclusion factor, particularly for reviews lacking a structured search strategy, clear inclusion criteria, or a transparent synthesis of findings. Studies that presented duplicated data without providing new insights were removed, as well as outdated reviews that had been superseded by more recent and comprehensive systematic reviews or meta-analyses.
After the research was carried out, all identified citations were compiled and entered into the Mendeley citation management system, with the systematic removal of duplicates.
Subsequently, the records were imported into the Rayyan QCRI reference manager [14]. This tool allows for fast and accurate processes, as it allows for the removal of duplicate studies, selection and screening of studies. In addition, it maintains methodological rigor and transparency among examiners, because it allows blind evaluation (Blind ON), thus avoiding possible biases [14].
In order to verify the agreement between the evaluators in the other selection phases, the Cohen’s Kappa Coefficient [15], presenting the following classification: 0-0.20, none; 0.21–0.39, minimal; 0.40–0.59, weak; 0.60–0.79, moderate; 0.80–0.90 strong; above 90, almost perfect. If there is agreement less than or equal to 0.79, training among the evaluators was carried out to increase the reliability of the process. For this, 30 studies were chosen to carry out the test with the evaluators.
Two review authors independently performed extract data using a pre-defined data extraction form. Where information from the reviews is unclear or missing, we will contact the review authors.
The extracted data from the included reviews covered general information such as authors, publication year, and the number of primary studies included. Population characteristics included demographics, average age, and prevalent health conditions like diabetes, cardiovascular diseases, Alzheimer’s, Parkinson’s, and depression. Intervention data detailed the types of probiotics used, including strains, dosages, frequencies, and administration methods (e.g., oral, capsules, or liquids). Comparators were analyzed based on the type of control group (placebo or standard treatment) and their characteristics. Cognitive outcomes were assessed using standardized cognitive function measures (e.g., MMSE, Montreal Cognitive Assessment), reporting improvements or declines. Metabolic outcomes included key indicators such as blood glucose, HbA1c, lipid profile, and blood pressure, evaluating the effects of probiotics on glycemic control, lipid metabolism, and overall metabolic health. A considerable for reviews without meta-analysis, a summary of the authors’ primary interpretation of the results was extracted. For meta-analyses, we extracted data on pooled effect sizes (e.g., rate ratio, risk ratio, odds ratio for dichotomous data, and mean difference or standardized mean difference for continuous data), as well as the corresponding 95% CIs and P values.
Systematic reviews exploring similar topics may have considerable overlap in the included primary studies. We created a citation matrix and calculate the corrected coverage area (CCA) index to analyze the overlap in the primary studies included in the reviews [16]. Based on the guidance from Hennessy and Johnson (2020) [17], we further examined the reasons for overlap based on the CCA value. Reviews with complete or near-complete overlap were analyzed to identify the causes of high overlap and considered for exclusion, prioritizing higher-quality studies (e.g. Cochrane reviews) and/or more recent reviews (if ratings are similar) were retained.
Study selection and assessment were conducted by two reviewers, using the JBI Critical Appraisal Checklist for Systematic Reviews and Research Synthesis. ‘Assessment of multiple systematic reviews’ (AMSTAR) [18]. The included reviews were descriptively synthesized and then, if feasible, the data were meta-analyzed.
The REML model was used as an estimator between the studies and a multivariate analysis was performed between the outcomes. It is a statistical method used to estimate parameters in variance-covariance models, especially in mixed linear models [19]. For stratification of the evidence, the number of studies, total number of participants, number of cases, p-value of the outcomes, inconsistency, imprecision, risk of bias and quality of the meta-analysis, prediction and the outcomes of each research were considered. The “ metaumbrella “ package was used, applying some specific tests for analysis, fixed-effect or random-effects meta-analysis, assessment of inconsistency/heterogeneity (I2), tests for effects of small studies and tests of excess statistical significance. If publication bias analysis is possible, it was demonstrated using the funnel plot graph, analyzed according to the trend of results and size of the study. The result of the inferential analysis was demonstrated using the forest plot. The R 4.3.2 software was used.
When summarising the findings of the reviews, we used the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) principles [20]for an overall assessment of the quality of evidence in the reviews for outcomes of interest. The quality of evidence for a given outcome was graded as high, moderate or low based on the overall quality of the systematic reviews and the risk of bias in primary studies, as well as the consistency of the results for an outcome.
RESULTS
Initially, 287 studies were found, of which, after applying the inclusion and exclusion criteria, 24 studies were selected.
The Systematic Review Citation Matrix (Figure 1) presented in the graph is a visual representation of the overlap of primary studies included in different systematic reviews. Each row and column correspond to a specific systematic review, and the blue filled squares indicate that a primary study was cited in both corresponding reviews. The scatter of points suggests a moderate level of overlap, evidenced by the fragmented pattern of markings, which indicates that some reviews share data sources, while others have separate studies. This analysis is useful for assessing redundancies or independence between reviews, helping to determine the reliability of the evidence and the need for possible exclusions to avoid duplication bias in meta-analyses.
Figure 2 presents the Forest Plot summarizing the effects of probiotics on cognitive and metabolic outcomes, based on a meta-analysis of several studies. The vertical axis lists the outcomes analyzed, including cognitive function, Alzheimer’s disease, Parkinson’s disease, depression, fasting glucose, HbA1c, lipid profile, blood pressure, triglycerides, and C-reactive protein (CRP). Each point on the plot represents the standardized mean effect size (SMD), while the horizontal bars indicate the 95% confidence interval (95% CI). The dashed line in the center marks the zero point, which represents the absence of a significant effect. If the confidence interval for an outcome crosses this line, it indicates that the results may not be statistically significant.
Additional information about each outcome is displayed on the right side of the graph. The “n-studies” column indicates how many studies were included to calculate the estimated effect of each variable. The “I2” column represents the heterogeneity of the studies, that is, the degree of variation between the results of the studies analyzed; higher values indicate greater variability between the studies. Finally, the “Class” classification categorizes the strength of the evidence based on the methodological quality and consistency of the results, ranging from Class II (moderate to high reliability) to Class IV (low reliability).
The analysis of the results indicates that probiotics have positive effects on cognitive function, reduction of inflammatory biomarkers (CRP) and metabolic control (HbA1c, glycemia, triglycerides and lipid profile). The effect on depression and Parkinson’s, although positive, has greater variability and uncertainty, reflected in the wide range of the 95% CI. In addition, the outcomes with high heterogeneity values (I2) suggest that the included studies have methodological or population differences, which may influence the reproducibility of the findings. Overall, this Forest Plot highlights the importance of probiotics as potential therapeutic interventions, but also highlights the need for more rigorous clinical trials to confirm their benefits in different health conditions.
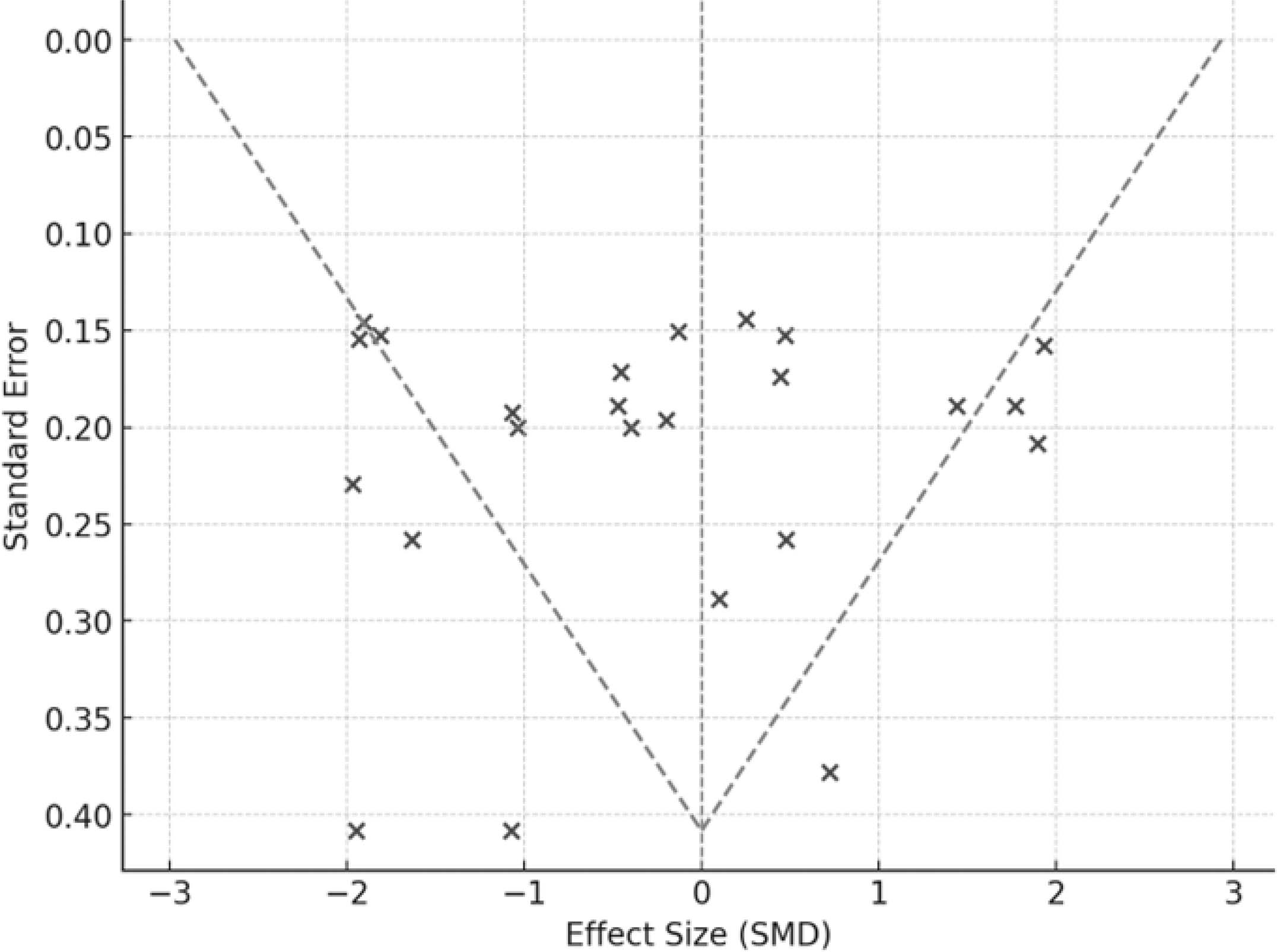
In the generated Funnel Plot (Figure 3), it is observed that some points are distributed asymmetrically, which may suggest publication bias or variability between studies. The presence of smaller studies with large positive effects and the absence of smaller studies with negative effects may indicate that non-significant results were less reported.
The sensitivity analysis (Figure 4) assesses how the mean effect size (MDS) changes when each study is individually excluded from the meta-analysis. Each blue dot represents the recalculated effect size when a specific study is removed. The gray dashed line represents the overall mean effect across all studies. If removing a study significantly changes the effect size, this suggests that the study has a strong influence on the results of the meta-analysis. In this case, although there are some fluctuations, no single study dramatically changes the overall effect, indicating moderate robustness of the findings.
The Evidence Map (Figure 5) shows the impact of probiotics on different health outcomes, organized into six categories: Cognitive Health, Mental Health, Metabolic Health, Inflammatory Response, Gut Health, and Cardiovascular Health. Each bubble represents an outcome assessed, where the size indicates the strength of the evidence (number of studies and robustness of findings), and the color reflects the level of certainty, with green for high certainty, yellow for moderate certainty, and orange for low certainty. It is observed that cognitive outcomes, such as cognitive function, Alzheimer’s, and Parkinson’s, have moderate to high evidence, suggesting potential benefits of probiotics in these aspects. In metabolic health, there is mixed evidence, with strong support for impacts on glycemic indexes, insulin resistance, and lipid composition, but with some uncertainties, especially in glucose control and glycated hemoglobin (HbA1c). The effects on the inflammatory response and gut health present robust evidence, indicating that probiotics can help reduce inflammatory markers and microbiota composition. In cardiovascular health, the findings vary, with greater certainty for effects on LDL and HDL cholesterol, but still some uncertainty for general markers of cardiovascular risk. Overall, this chart highlights areas where probiotics have strong scientific support and where there are still gaps in the research, and is a useful tool to direct future investigations and guide evidence-based clinical decisions.
The table 2 presents an assessment of the certainty of evidence regarding the metabolic and cardiovascular effects of probiotics compared to standard treatment, with an average follow-up of 6 months. It includes 15 systematic reviews of randomized clinical trials, with no serious risk of bias, inconsistency, indirect evidence, or inaccuracy. The sample consisted of 9,720 patients in the probiotics group and 9,734 in the control group. The relative effect suggests a 75% increase in the analyzed outcomes (95% CI: 64 to 80). The certainty of the evidence was rated as high (++++), indicating reliable results with critical clinical importance.
Table 2 : Assessment of methodological quality using the GRADE system (Metabolic and cardiovascular effects).
| Certainty assessment | No. of patients | Effect | Certainty | Importance | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of studies | Study design | Risk of bias | Inconsistency | Indirect evidence | Inaccuracy | Other considerations | Probiotics | Standard treatment | Relative (95% CI) | Absolute (95% CI) | ||
| Metabolic and cardiovascular effects (follow-up: average 6 months) | ||||||||||||
| 15 | Systematic reviews - randomized clinical trials | do not serious | do not serious | do not serious | do not serious | none | 9720 | 9734 | - | 75% higher (64 higher to 80 higher) | High ++++ | Critical |
CI: Confidence interval
The table 3 presents an assessment of the certainty of evidence regarding the cognitive effects of probiotics compared to standard treatment, with an average follow-up of 6 months. It includes 9 systematic reviews of randomized clinical trials, with no serious risk of bias, inconsistency, indirect evidence, or inaccuracy. The sample consisted of 3,650 patients in the probiotics group and 3,657 in the control group. The relative effect suggests a 61% increase in the analyzed outcomes (95% CI: 58 to 78). The certainty of the evidence was rated as high (++++), indicating reliable results with critical clinical importance.
Table 3 : Assessment of methodological quality using the GRADE system (Metabolic and cardiovascular effects).
| Certainty assessment | No. of patients | Effect | Certainty | Importance | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of studies | Study design | Risk of bias | Inconsistency | Indirect evidence | Inaccuracy | Other considerations | Probiotics | Standard treatment | Relative (95 %CI) | Absolute (95% CI) | ||
| Cognitive effects (follow-up: average 6 months) | ||||||||||||
| 9 | Systematic reviews - randomized clinical trials | do not serious | do not serious | do not serious | do not serious | none | 3650 | 3657 | - | 61% higher (58 higher to 78 higher) | ++++ High | Critical |
CI: Confidence interval
Table 1 : Summary of Included Systematic Reviews and Meta-Analyses on the Cognitive and Metabolic Effects of Probiotic Supplementation in Older Adults.
| Authors | Number of Studies Included | Total Sample | Participant Characteristics | Evaluated Intervention | Comparison | Outcomes Evaluated | Main Results | Authors' Conclusions | Strength of Evidence | Strain(s) Evaluated | Avaliação AMSTAR |
|---|---|---|---|---|---|---|---|---|---|---|---|
| NASERI et al. (2023) | 32 | 2074 participants | Individuals with prediabetes and type 2 diabetes (T2DM), age between 46.4 and 66 years, BMI between 22.4 and 35.6 kg/m2 | Probiotic and synbiotic supplementation (various strains and forms: capsules, powder, yogurt, fermented milk) | Placebo or usual care | CRP, TNF-α, IL-6, GSH, MDA, TAC, NO, Adiponectin, Leptin | Significant reduction of CRP (-0.62 mg/L), TNF-α (-0.27 pg/mL) and MDA (-0.51 µmol/L); Significant increase of GSH (+69.80 µmol/L), TAC (+73.59 mmol/L) and NO (+7.49 µmol/L); No significant changes in IL-6, adiponectin and leptin | Supplementation with probiotics or synbiotics may reduce inflammation and oxidative stress in patients with prediabetes and type 2 diabetes, potentially reducing the risk of cardiovascular disease. | Low to very low (assessed by GRADE method) | Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus reuteri, Bifidobacterium animalis, Lactobacillus sporogenes, Saccharomyces cerevisiae | Moderada |
| GHORBANI et al. (2023) | 40 | 2795 participants | Patients with cardiometabolic risk factors including dyslipidemia, type 2 diabetes, metabolic syndrome, obesity, and NAFLD | Probiotic and synbiotic supplementation (various strains and forms: capsules, powder, sachets, tablets) | Placebo | Triglycerides, Total Cholesterol, LDL-C, HDL-C | Significant reduction in triglycerides (-12.26 mg/dL), total cholesterol (-8.43 mg/dL) and LDL-C (-5.08 mg/dL). Slight increase in HDL-C (+1.14 mg/dL) | Supplementation with probiotics and synbiotics can improve dyslipidemia, with greater impact in individuals with hypercholesterolemia and metabolic syndrome | Variable (heterogeneity between studies), with a need for more high-quality RCTs | Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus rhamnosus, Lactobacillus plantarum, Lactobacillus reuteri, Bifidobacterium bifidum, Bifidobacterium breve, Bifidobacterium lactis, Bifidobacterium longum, Streptococcus thermophilus | Moderada |
| LIU et al. (2023) | 10 | 715 participants | Individuals with cognitive impairment and Alzheimer's, age 50 to 100 years | Probiotic supplementation (different strains and doses, administered via capsules, fermented milk or sachets) | Placebo | Cognitive function (Mini-Mental State Examination - MMSE, RBANS, ADAS-Jcog, NIH Toolbox) | Significant improvement in cognitive function in individuals with Alzheimer's or cognitive impairment (SMD = 1.34; 95% CI, 0.51–2.16; P < 0.01) | Probiotic supplementation showed a significant positive effect on cognition in individuals with cognitive impairment, but no effect in healthy individuals | Moderate to high for individuals with Alzheimer's and cognitive impairment; low for individuals without a diagnosis | Lactobacillus acidophilus, Lactobacillus casei, Bifidobacterium bifidum, Lactobacillus fermentum, Bifidobacterium longum BB536, Bifidobacterium infantis M-63, Bifidobacterium breve MCC1274, Bifidobacterium breve M-16V, Bifidobacterium breve A1, Lactobacillus helveticus, Lactobacillus rhamnosus GG, Enterococcus faecalis | Alta |
| HONG et al. (2022) | 8 | 560 participants | Patients with Parkinson's disease and gastrointestinal symptoms, age 61 to 75 years | Probiotic supplementation (different strains and doses, administered via capsules, fermented milk or sachets) | Placebo | Bowel movements, stool consistency, systemic inflammation, metabolism | Significant increase in bowel movement frequency (MD: 1.06/week; 95% CI, 0.61–1.51; p < 0.001) and normalization of stool consistency (SMD: 0.61; 95% CI, 0.31–0.91; p < 0.001) | Probiotic supplementation significantly improved constipation in Parkinson's patients and may have systemic effects on inflammation and metabolism | Moderate (variability between studies, need for more long-term RCTs) | Lactobacillus acidophilus, Lactobacillus casei, Bifidobacterium bifidum, Lactobacillus plantarum, Bifidobacterium breve, Enterococcus faecalis, Streptococcus thermophilus, Limosilactobacillus | Alta |
| SAMAH et al. (2016) | 6 | 980 participants | Patients with Type 2 Diabetes Mellitus | Probiotic supplementation | Placebo | Fasting blood glucose (FBG), Glycated hemoglobin (HbA1c), Fasting plasma insulin, HOMA-IR, C-reactive protein, IL-6, Malondialdehyde | Significant reduction in fasting glucose (MD = -0.98 mmol/L; 95% CI, -1.17 to -0.78; p < 0.00001). There were no significant differences for HbA1c and other secondary outcomes | Moderate hypoglycemic effect of probiotics, significantly reducing fasting blood glucose. However, effects on HbA1c, inflammation and oxidative stress remain inconsistent | Moderate (need for more well-designed studies) | Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus reuteri, Bifidobacterium animalis, Lactobacillus sporogenes, Saccharomyces cerevisiae | Moderada |
| KRÜGER et al. (2020) | 3 | 161 participants | Individuals with Alzheimer's Disease | Probiotic supplementation (Lactobacillus and Bifidobacterium strains) | Placebo | Cognitive function, plasma triglycerides, VLDL cholesterol, insulin resistance, plasma malondialdehyde | No significant effect on cognitive function (SMD = 0.56; 95% CI, -0.06 to 1.18), but improvement in triglycerides, VLDL cholesterol, insulin resistance, and plasma malondialdehyde | Insufficient evidence to recommend clinical use of probiotics in individuals with dementia. Improvement observed in some metabolic and inflammatory biomarkers | Very low (assessed by the GRADE method) | Lactobacillus spp., Bifidobacterium spp. | |
| RUIZ-GONZALEZ et al. (2020) | 16 | 360 participants | Individuals with dementia and mild cognitive impairment (MCI) | Probiotic supplementation (Lactobacillus and Bifidobacterium) | Placebo | General cognitive function, memory, spatial and non- spatial learning, brain-derived neurotrophic factor (BDNF), inflammatory profile, cellular biomarkers | Improvement in general cognitive function in humans after probiotic supplementation; improvement in memory and spatial learning in animal models; increase in BDNF levels and improvement in the inflammatory profile | Probiotic supplementation may be a promising therapeutic strategy for dementia and mild cognitive impairment, but more clinical studies are needed to strengthen the evidence | Moderate for preclinical, limited for clinical | Lactobacillus spp., Bifidobacterium spp. | Alta |
| ZHANG et al. (2021) | 33 | 1927 participants | Adult patients with Type 2 Diabetes Mellitus | Probiotic supplementation (doses ≥ 10⁹ CFU/day, including single and multiple strains) | Placebo | Glycated hemoglobin (HbA1c), fasting glucose, fasting insulin, HOMA-IR | Significant reduction in HbA1c (MD: -0.19%; 95% CI, -0.32 to -0.07; p = 0.003), fasting plasma glucose (MD: -1.00 mmol/L; 95% CI, -1.45 to -0.56; p < 0.0001), fasting insulin (MD: -5.73 pmol/L; 95% CI, -12.17 to 0.72; p = 0.08) and HOMA-IR (MD: -1.00; 95% CI, -1.32 to -0.68; p < 0.00001) | Probiotic supplementation leads to modest reductions in fasting glucose and fasting insulin. High-dose, multistrain probiotics appear to have the greatest impact on glycemic control | Low for HbA1c and fasting glucose, moderate for fasting insulin, high for HOMA-IR | Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus reuteri, Bifidobacterium animalis, Lactobacillus sporogenes, Saccharomyces cerevisiae | Alta |
| LIU et al. (2023) | 6 | 512 participants | Adults at cardiovascular risk, including hypercholesterolemia and metabolic syndrome | Limosilactobacillus reuteri supplementation (varying doses and different strains) | Placebo | Systolic and diastolic blood pressure, fasting blood glucose, total cholesterol (TC), LDL-C, HDL-C, triglycerides (TG) | Significant reduction in total cholesterol (TC) (-0.26 mmol/L; 95% CI, -0.48 to -0.03; p = 0.02). No significant effect on blood pressure, LDL-C, HDL-C, TG or fasting glucose | Consumption of L. reuteri significantly reduces total cholesterol levels, especially in individuals <55 years old, with a BMI between 25-30 or with hypercholesterolemia. There was no significant impact on other metabolic factors | Moderate for total cholesterol reduction, low for other outcomes | Limosilactobacillus reuteri NCIMB 30242, ADR-1, V3401, DSM 17938 | Alta |
| PERNA et al. (2021) | 20 | 1411 participants | Overweight and obese adults with associated metabolic diseases | Probiotic supplementation (different strains and doses) | Placebo | Body weight, BMI, waist circumference (WC), hip circumference (HC) | No significant effect on weight reduction (-0.26 kg; 95% CI, -0.75 to 0.23; p = 0.30); Significant reduction in BMI (-0.73 kg/m2; 95% CI, -1.31 to -0.16; p = 0.01), WC (-0.71 cm; 95% CI, -1.24 to -0.19; p = 0.008) and HC (-0.73 cm; 95% CI, -1.16 to -0.30; p = 0.0008) | Probiotic supplementation showed positive effects on reducing BMI, WC and HC, but not on weight loss. Future studies should evaluate the efficacy of different strains, doses and duration of intervention | Moderate for BMI, WC and HC; low for body weight | Lactobacillus gasseri, Lactobacillus rhamnosus, Lactobacillus plantarum, Bifidobacterium infantis, Bifidobacterium longum, Bifidobacterium breve B3 | Moderada |
| CHENHUICHEN et al. (2022) | 10 | 475 participants | Individuals aged 65 and over | Probiotic and prebiotic supplementation | Placebo | Glucose homeostasis, cognitive function, frailty phenotype, gut microbiota profile, immunological parameters | Significant improvement in parameters such as glucose homeostasis, cognition and immune profile; some parameters remained unchanged | Probiotics and prebiotics have potential to modulate aging and prevent health problems in the elderly, but more studies are needed | Moderate for some outcomes, low for others due to heterogeneity of studies | Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus reuteri, Bifidobacterium animalis, Lactobacillus sporogenes, Saccharomyces cerevisiae | Alta |
| CUSTODERO et al. (2018) | 49 | Average of 210 participants per study | Middle-aged and older adults with chronic low-grade inflammation | Supplementation with probiotics and other nutritional and pharmacological compounds | Placebo | Interleukin-6 (IL-6) and C-Reactive Protein (CRP) Levels | Significant reduction in IL-6 (-0.68 pg/ml) and CRP (-0.43 mg/L) levels after probiotic supplementation | Probiotics and some other compounds may reduce inflammatory markers in older adults, but there is high heterogeneity in studies | Moderate for reduction of IL-6 and CRP | Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus reuteri, | Alta |
| JIANG et al. (2020) | 25 | 3 randomized clinical trials (173 patients) and 22 preclinical studies | Multiple Sclerosis (MS) patients and animal models of the disease | Probiotic supplementation | Placebo | Mental health (EDSS, BDI, GHQ, DASS scales), insulin resistance, inflammatory and oxidative stress markers | Significant improvement in mental health (EDSS: SMD = -1.22; BDI: SMD = -1.58; GHQ: SMD = -0.71; DASS: SMD = -0.72, p < 0.05). Probiotics also reduced insulin resistance, inflammation and oxidative stress | Probiotics may have beneficial effects in the prevention and treatment of Multiple Sclerosis, but the certainty of the evidence is very low due to the high heterogeneity of the studies | Very low due to high heterogeneity | Lactobacillus paracasei, | Alta |
| DING et al. (2021) | 17 | 836 participants | Adults with Type 2 Diabetes Mellitus | Probiotic supplementation | Placebo | Inflammatory markers (TNF-α, CRP, IL-6), fasting glucose (FPG), glycated hemoglobin (HbA1c), HOMA-IR | Significant reduction in TNF-α (-0.37; 95% CI, -0.56 to -0.19; p < 0.0001) and CRP (-0.21; 95% CI, -0.42 to -0.01; p = 0.040); No significant effect on IL-6. Improved glycemic control with reduction in FPG, HbA1c and HOMA-IR | Probiotic supplementation may improve glucose homeostasis and reduce inflammatory markers in individuals with T2DM. However, further studies are needed to confirm these effects. | Moderate for TNF-α and CRP, low for IL-6 and blood glucose | Not specified | Alta |
| PONTES et al. (2021) | 26 | 1720 participants | Overweight and obese individuals | Probiotic supplementation (doses ≥ 101⁰ CFU/day, duration ≥ 8 weeks) | Placebo | Body weight, BMI, waist circumference (WC), fat mass, TNF-α, insulin, total cholesterol, LDL | Significant reduction in body weight (-0.70 kg; 95% CI, -1.04 to -0.35 kg; p < 0.0001), BMI (-0.24 kg/m2; 95% CI, -0.35 to -0.12; p = 0.0001), WC (-1.13 cm; 95% CI, -1.54 to -0.73 cm; p < 0.0001), fat mass (-0.71 kg; 95% CI, -1.10 to -0.32 kg; p = 0.0004), TNF-α (-0.16 pg/ml; 95% CI, -0.24 to -0.08 pg/ml; p = 0.0001), insulin (-0.85 mcU/ml; 95% CI, -1.50 to -0.21 mcU/ml; p = 0.010), total cholesterol (-0.16 mmol/l; 95% CI, -0.26 to -0.05 mmol/l; p = 0.003), LDL (-0.09 mmol/l; 95% CI, -0.16 to -0.03 mmol/l; p = 0.006) | Probiotic supplementation may be useful for improving body weight, adiposity and some cardiovascular risk markers in overweight and obese individuals | Moderate for adiposity and metabolic markers | Lactobacillus acidophilus | Alta |
| SUN & BUYS (2015) | 15 | 788 participants | Individuals with cardiovascular risk factors | Probiotic supplementation (fermented milk or capsules, consumption ≥ 8 weeks) | Placebo | Total cholesterol, LDL, BMI, waist circumference, inflammatory markers (CRP, TNF-α) | Significant reduction in total cholesterol (-0.27 mmol/L; 95% CI, -0.38 to -0.16; p < 0.00001) and LDL (-0.23 mmol/L; 95% CI, -0.33 to -0.13; p < 0.00001). Moderate reduction in BMI (-0.52 kg/m2) and waist circumference (-2.11 cm) | Probiotics are effective in reducing lipids and inflammatory markers, especially when consumed in fermented products for ≥ 8 weeks and containing multiple strains | Moderate for total and LDL cholesterol, low for other outcomes | Lactobacillus acidophilus, Streptococcus thermophilus, Bifidobacterium spp., Lactobacillus plantarum, Enterococcus spp. | Alta |
| GUTIERREZ et al. (2021) | 61 | 1300 participants | Adults with or without cognitive impairment | Dietary interventions, nutritional supplementation and eating patterns | Usual diet or placebo | General cognitive function, brain integrity, biomarkers of inflammation and oxidative stress | Reasonable evidence that dietary patterns, foods, and supplements improve cognitive domains or measures of brain integrity. Mediterranean diet has shown promising results, while the efficacy of DASH diet is unclear | Healthy eating patterns and certain supplements may help prevent cognitive decline, but heterogeneity of studies limits definitive conclusions | Variable (moderate for polyphenols and nutrient combinations, low for PUFAs and vitamin D) | Lactobacillus acidophilus | Moderada |
| HUANG et al. (2016) | 5 | 365 participants | Healthy individuals and patients with major depression | Probiotic supplementation | Placebo | Depression scales (DASS, BDI, HADS-D, POMS) | Significant reduction in depression scales (MD = -0.30; 95% CI, -0.51 to -0.09; p = 0.005). Greater effect in patients with major depression (MD = -0.73; 95% CI, -1.37 to -0.09; p = 0.03) | Probiotics have been associated with a significant reduction in symptoms of depression, especially in individuals with major depressive disorder | Low to moderate | Lactobacillus acidophilus, Lactobacillus casei, Bifidobacterium bifidum, Lactobacillus helveticus, Bifidobacterium longum, Lactobacillus rhamnosus, Streptococcus thermophilus | Moderada |
| NIKBAKHT et al. (2016) | 14 | 180 participants | Adults with varying blood glucose levels | Probiotic and symbiotic supplementation | Placebo | Fasting blood glucose (FBG) | Borderline significant overall reduction in fasting glucose (-0.18 mmol/L; 95% CI, -0.37 to 0.00; p = 0.05). More pronounced reduction in individuals with FBG ≥7 mmol/L (-0.68 mmol/L; 95% CI, -1.07 to -0.29; p < 0.01). Multispecies probiotics were more effective (-0.31 mmol/L; 95% CI, -0.58 to -0.03; p = 0.03) than single-species probiotics | Probiotics and synbiotics may be beneficial in reducing fasting glucose, especially in individuals with elevated FBG. Multispecies probiotics appear to have the greatest impact | Moderate reduction in fasting glucose in individuals with FBG ≥7 mmol/L | Lactobacillus acidophilus | |
| EJTAHED et al. (2020) | 5 | 2703 participants | Normotensive adults, hypertensive adults, adults with type 2 diabetes (T2DM) or metabolic syndrome | Probiotic supplementation (different strains, fermented foods, supplements) | Placebo | Systolic blood pressure (SBP), diastolic blood pressure (DBP) | Moderate reduction in systolic blood pressure (-3.10 to -5.04 mmHg) and diastolic blood pressure (-0.39 to -3.84 mmHg). Effects more evident in individuals with blood pressure ≥ 130/85, in Asian fermented products and in multispecies probiotics with doses ≥ 1011 CFU | High-dose multispecies probiotics may be effective in controlling blood pressure, especially in hypertensive individuals | Moderate for reducing blood pressure in hypertensive individuals | Lactobacillus spp., Bifidobacterium spp., Enterococcus spp., Streptococcus spp., Saccharomyces cerevisiae | Moderada |
| MOSQUERA et al. (2024) | 51 | 3353 participants | Adults with psychiatric and cognitive disorders, especially depression, bipolar disorder, schizophrenia, and cognitive impairment | Psychobiotic supplementation (probiotics that act on the gut-brain axis) | Placebo | Psychiatric symptoms (depression, anxiety, schizophrenia, bipolar disorder), cognitive function, inflammatory biomarkers, neurotransmitters | Significant improvement in depressive and cognitive symptoms, especially in interventions with Lactobacillus and Bifidobacterium. High variability between studies makes definitive conclusions difficult | Psychobiotics show therapeutic potential in the management of psychiatric and cognitive disorders, but further studies are needed to optimize strains, dosage and duration of treatment | Moderate for depression, low for schizophrenia and bipolar disorder due to heterogeneity of studies | Lactobacillus spp., Bifidobacterium spp., Streptococcus thermophilus, Enterococcus spp. | Alta |
| LIVESEY et al. (2019) | 24 | Several prospective cohort studies | Initially healthy adults of diverse ethnicities | Glycemic index (GI) and glycemic load (GL) of the diet | Diets with lower GI and GL | Relationship between GI, GL and risk of type 2 diabetes | Relative risk of developing type 2 diabetes increased with high GI (RR = 1.27, 95% CI: 1.15–1.40, p < 0.001) and high GL (RR = 1.26, 95% CI: 1.15–1.37, p < 0.001) diets. Effect was more pronounced in populations of European and East Asian descent | Diets with high GI and GL are associated with an increased risk of developing type 2 diabetes. It is recommended that these factors be considered in nutritional guidelines | High GI and GL as risk factors for type 2 diabetes | Lactobacillus acidophilus | Moderada |
| LIANG et al. (2022) | 25 | 2843 participants | Patients with type 2 diabetes | Probiotic supplementation and comparison with glucose-lowering drugs (GLP-1 RA, SGLT-2i, TZD, DPP-4i) | Placebo and different classes of hypoglycemic drugs | Fasting blood glucose (FBS), glycated hemoglobin (HbA1c), total cholesterol (TC), triglycerides (TG), systolic blood pressure (SBP) and diastolic blood pressure (DBP) | Multispecies probiotics significantly reduced FBS, TC, SBP and DBP. Stronger effect was observed in patients aged >55 years, BMI ≥30 kg/m2 and prolonged intervention duration | Multispecies probiotics may be more beneficial than hypoglycemic drugs in preventing type 2 diabetes in patients with multiple comorbidities, but more studies are needed | Moderate for glycemic and lipid reduction, low for impact on HbA1c | Lactobacillus acidophilus | Moderada |
| VAGHEF-MEHRABANY et al. (2020) | 32 | 320 participants | Healthy adults and patients with psychiatric disorders, including depression, anxiety, and bipolar disorder | Psychobiotic supplementation (probiotics, prebiotics and synbiotics) | Placebo | Symptoms of depression (assessed by BDI, HADS, HAM-D, DASS), inflammatory biomarkers, neuroendocrine axes | Only seven studies reported significant antidepressant effects of psychobiotics. Inconsistent results between studies, with variable effect depending on the probiotic strain used | Psychobiotics may impact depressive symptoms in a strain-dependent manner, but the evidence is still inconclusive. Future studies should consider individual microbiota variations and specific depression subtypes. | Low to moderate due to high heterogeneity of studies | Lactobacillus spp., Bifidobacterium spp., Streptococcus thermophilus, Clostridium butyricum, Bacillus coagulans |
DISCUSSION
The results of this review reinforce the growing evidence on the beneficial effects of probiotics on cognitive function and metabolism in elderly individuals with chronic conditions such as diabetes mellitus, cardiovascular disease, Alzheimer’s, Parkinson’s and depression. The role of probiotics in modulating the intestinal microbiota and their influence on the gut-brain axis has been increasingly studied, demonstrating their ability to alter inflammatory processes, regulate oxidative stress and neurotransmission. These findings support the hypothesis that the composition of the intestinal microbiota can directly influence the functioning of the central nervous system, impacting aspects such as memory, cognition and mood. However, despite the promising evidence, the included studies showed significant heterogeneity in terms of type of probiotic used, intervention time and characteristics of the studied population, which reinforces the need for more homogeneous and well-controlled studies for a more precise understanding of the mechanisms involved.
The relationship between gut microbiota and cognition has been widely discussed in the scientific literature, and the results of this meta-umbrella indicate that probiotic supplementation may play a neuroprotective role in the elderly [21], especially those with neurodegenerative diseases. The central hypothesis suggests that probiotics, by promoting a more balanced intestinal environment, favor the production of short-chain fatty acids, which are essential for maintaining the blood-brain barrier and regulating neuroinflammation [22]. Furthermore, the regulation of the gut microbiota directly influences the release of neurotransmitters, such as serotonin and gamma-aminobutyric acid (GABA), which play fundamental roles in regulating mood and cognitive functions [23]. Thus, the relationship between probiotics and cognition can be explained by a complex interaction between inflammatory, metabolic, and neurotransmitter factors, as evidenced in the findings of this review.
The improvement in metabolic outcomes observed with the use of probiotics can be explained by their role in modulating glucose and lipid metabolism, determining factors for maintaining metabolic homeostasis in elderly individuals with chronic conditions. Studies suggest that probiotic supplementation can improve insulin sensitivity, reduce peripheral resistance, and promote better glycemic regulation in patients with type 2 diabetes [24]. This effect may be associated with the ability of probiotics to reduce low-grade chronic inflammation, which is one of the main characteristics of metabolic diseases [25]. Furthermore, the influence of probiotics on the modulation of the intestinal microbiota may favor the production of metabolites that regulate lipid metabolism, helping to reduce total cholesterol and triglycerides, essential factors for the prevention of cardiovascular diseases [26].
Metabolic syndrome (MS) has increasingly been recognized as a significant risk factor for chronic non-communicable diseases, such as type 2 diabetes and cardiovascular diseases, particularly in vulnerable populations. A cross-sectional study conducted with artisanal fishermen in Tocantins identified a high prevalence of MS, with abdominal obesity being the most prevalent component. Additionally, factors such as smoking and socioeconomic status showed a significant association with the presence of the syndrome, indicating that social determinants of health may directly influence metabolic risk in this population[27].
The relationship between probiotics and metabolism is supported by several physiological mechanisms, including the reduction of intestinal permeability and the modulation of the systemic inflammatory response. Intestinal dysbiosis, a condition frequently observed in the elderly [28]and individuals with chronic diseases, is associated with an increase in intestinal permeability, allowing the translocation of endotoxins into the systemic circulation, resulting in chronic inflammation [29]. Probiotics help maintain the integrity of the intestinal barrier, preventing this phenomenon and reducing the levels of inflammatory biomarkers, such as interleukin-6 (IL-6) and tumor necrosis factor alpha (TNF-α). Thus, the restoration of intestinal homeostasis can have systemic effects that positively impact energy metabolism, resulting in better regulation of blood glucose and serum lipids [30].
Publication bias analysis indicated that smaller studies with positive results were more likely to be published, which may influence the interpretation of the findings. However, tests for excess statistical significance and funnel plots demonstrated that, despite the existence of publication bias in some reviews, the observed effects remain consistent within the most rigorous analyses. This reinforces the need for high-quality randomized clinical trials, with larger sample sizes and standardized methodologies, to confirm the effects of probiotics on the cognitive and metabolic health of older adults.
Variability in the study population also represents an important limitation. The presence of different comorbidities in the participants of the primary studies may have influenced the outcomes, since factors such as systemic inflammation, medication use and nutritional status can interfere with the response to probiotics [31]. Future studies should seek more detailed subanalyses to determine whether the effects of probiotics vary according to the clinical profile of patients, taking into account factors such as degree of cognitive impairment, metabolic status and composition of the basal intestinal microbiota [32].
The duration of probiotic treatment has also been shown to be a determining factor in obtaining significant benefits. While some studies have reported improvements within a few weeks [33], others have indicated that positive effects only appear after months of continuous supplementation [34]. This suggests that stabilization of the intestinal microbiota may be a gradual process, requiring prolonged interventions to obtain lasting clinical benefits. Thus, defining the ideal duration of supplementation still represents a challenge to be clarified in future studies [35].
The clinical applicability of the findings of this meta-umbrella is a crucial aspect to be considered. Probiotic supplementation stands out as an accessible and low-cost intervention that can be easily incorporated into care routines in older adults, especially those with chronic diseases. However, for its effective implementation, it is essential that health professionals consider the individualization of treatment, taking into account the patient’s characteristics, the choice of the most appropriate probiotic strain and the association with other nutritional and therapeutic interventions [36].
Increasing evidence suggests that the gut microbiota may influence Parkinson’s pathogenesis through the gut-brain axis, a bidirectional pathway connecting the central nervous system to the gastrointestinal tract [37]. Parkinson’s patients often present with gut dysbiosis, characterized by a reduction in the diversity of beneficial bacteria and an increase in the abundance of pro-inflammatory microorganisms [38]. This alteration is associated with a state of chronic inflammation, which may exacerbate neurodegeneration and disease progression [39]. Studies included in the present review suggest that probiotic supplementation may help restore the gut microbiota, reducing the production of inflammatory cytokines and promoting the synthesis of neuroprotective metabolites, such as short-chain fatty acids [40]. Furthermore, there is evidence that probiotics may improve non-motor symptoms of Parkinson’s, such as gastrointestinal disturbances and mood alterations, both of which are strongly correlated with disease progression [41].
In the case of Alzheimer’s, the results of this review reinforce the role of the intestinal microbiota in the pathophysiology of the disease, which is characterized by the abnormal accumulation of beta-amyloid plaques and neurofibrillary tangles in the brain [42]. Neuroinflammation has been identified as a determining factor in the progression of Alzheimer’s, and there is growing interest in understanding how the intestinal microbiota can influence this process. Patients with Alzheimer’s often present an altered intestinal microbiota profile, with an increase in pathogenic bacteria that produce lipopolysaccharides (LPS), which can cross the intestinal barrier and trigger systemic inflammation and neuroinflammation [43]. The reviewed studies suggest that probiotic supplementation can modulate this inflammatory environment, reducing LPS production and promoting the integrity of the blood-brain barrier, thus reducing neurotoxicity [44]. Furthermore, probiotics can positively influence the synthesis of neurotransmitters and neurotrophic factors essential for the maintenance of synaptic plasticity, resulting in improved cognition and reduced neurodegenerative decline [45].
Depression in the elderly is a multifactorial condition that may be influenced by the composition of the gut microbiota, considering that the gut houses most of the serotonin-producing cells in the human body [46]. Dysregulation of the gut-brain axis has been associated with a reduction in the availability of neurotransmitters such as serotonin and dopamine, as well as an increase in the systemic inflammatory response, both central factors in the pathophysiology of depression [47]. The results of this review indicate that probiotic supplementation may act in mood regulation, promoting the production of bioactive metabolites with antidepressant action and reducing systemic inflammation [48]. Furthermore, there is evidence that probiotics may modulate the hypothalamic-pituitary-adrenal (HPA) axis, reducing cortisol levels and attenuating the stress response, a key factor in the persistence of depressive symptoms [49]. Therefore, the use of probiotics may represent a promising complementary strategy for the management of geriatric depression, reducing the need for isolated pharmacological interventions [50].
The relationship between anxiety and the gut microbiota has also been widely studied, since changes in the microbiota can directly affect the stress response and emotional regulation [51]. The studies included in this review suggest that probiotics may influence the reduction of anxiety symptoms through modulation of the inflammatory response and increased bioavailability of inhibitory neurotransmitters, such as gamma-aminobutyric acid (GABA) [52]. Furthermore, the reduction in intestinal inflammation promoted by probiotics may improve the integrity of the blood-brain barrier, reducing neuronal hyperactivity associated with anxiety disorders [53]. Another proposed mechanism involves the modulation of the HPA axis, decreasing cortisol release and promoting a more balanced response to stress [54]. Although the evidence is promising, there is a need for more clinical studies evaluating the impact of specific strains on reducing anxiety in older adults, considering individual differences in microbiota composition and inflammatory profiles [55].
In the context of metabolic diseases, probiotics have demonstrated significant potential in regulating glucose and lipid metabolism, contributing to improving the metabolic profile in elderly individuals with chronic conditions [56]. Gut dysbiosis has been associated with increased insulin resistance, systemic inflammation, and impaired energy metabolism, factors that contribute to the development of metabolic diseases. The results of this review indicate that probiotic supplementation can improve glycemic homeostasis, promoting better insulin sensitivity and reducing the production of inflammatory mediators such as IL-6 and TNF-α. Furthermore, regulation of the intestinal microbiota can directly influence nutrient absorption and the production of metabolites essential for energy balance [57], resulting in better modulation of lipid metabolism and fat deposition [58].
In the specific case of type 2 diabetes mellitus, the findings reinforce the importance of the intestinal microbiota in the regulation of glycemia and insulin resistance [59]. The intestinal dysbiosis observed in diabetic patients is characterized by increased intestinal permeability and endotoxin translocation, triggering a systemic inflammatory response that aggravates insulin resistance [60]. Probiotic supplementation has been shown to reduce these inflammatory processes, restoring the integrity of the intestinal barrier and promoting better glucose uptake by peripheral tissues. Furthermore, some specific probiotic strains have been associated with the production of incretins, gastrointestinal hormones that improve insulin secretion and glycemic regulation. These findings suggest that probiotics may be an adjuvant therapeutic strategy in the management of diabetes, helping to reduce fasting glycemia and glycated hemoglobin (HbA1c) [61].
Cardiovascular diseases, in turn, are directly related to the individual’s metabolic and inflammatory profile, and the intestinal microbiota plays a fundamental role in this context [62]. Intestinal dysbiosis has been associated with increased production of trimethylamine-N-oxide (TMAO), a metabolite that contributes to the development of atherosclerosis and cardiovascular events [63]. Probiotic supplementation can modulate TMAO production [64]and reduce LDL cholesterol and triglyceride levels, promoting a more balanced lipid profile and reducing the risk of cardiovascular complications [65]. Furthermore, there is evidence that probiotics can exert hypotensive effects by regulating nitric oxide bioavailability and improving endothelial function, essential factors for vascular health in the elderly [66].
Another relevant aspect to be discussed is the interaction between the intestinal microbiota and obesity, a condition frequently associated with unfavorable metabolic outcomes. Regulation of the intestinal microbiota through probiotic supplementation may influence energy homeostasis, reducing the absorption of short-chain fatty acids and promoting more efficient lipid metabolism. Furthermore, some probiotic strains have demonstrated the ability to modulate the expression of genes related to thermogenesis and energy expenditure, suggesting a possible impact on reducing visceral adiposity and improving body composition in overweight elderly individuals [67].
This umbrella review provides a comprehensive synthesis of existing evidence on the cognitive and metabolic effects of probiotics in older adults while offering new insights into the patterns, contradictions, and gaps that emerge from this extensive body of literature. Unlike individual systematic reviews and meta-analyses, which often focus on specific subgroups or outcomes, this study integrates findings from multiple sources to provide a more holistic understanding of the impact of probiotics. The review highlights how probiotic supplementation may influence cognitive function, metabolic parameters, and inflammatory markers, identifying the most effective probiotic strains and dosages. Additionally, it reveals inconsistencies in reported outcomes, emphasizing the need for standardized clinical trials to establish more conclusive recommendations. By systematically evaluating methodological quality, this study ensures that conclusions are based on the highest levels of evidence, reducing bias and enhancing the reliability of findings.
Beyond summarizing existing data, this review contributes by identifying key research gaps and guiding future investigations. One of its major strengths lies in mapping the heterogeneity of included studies, allowing for a nuanced discussion of why certain probiotics yield inconsistent results across different populations and health conditions. It also underscores the interaction between probiotics and host factors, such as gut microbiota composition, baseline metabolic health, and the presence of comorbidities, which may influence their efficacy. This review also provides a foundation for refining clinical guidelines, helping healthcare practitioners and policymakers make informed decisions about probiotic use in older adults. By identifying areas requiring further research, such as optimal intervention durations, specific probiotic strains, and their role in multi-targeted health interventions, this study plays a crucial role in advancing the scientific understanding of probiotics as a therapeutic strategy for aging-related conditions.
The results of this review also highlight the need for clearer guidelines on the use of probiotics in older adults. Currently, there is no consensus on the optimal dose, the best combination of strains, and the duration of treatment for different health conditions. Standardizing these variables could facilitate prescription and clinical monitoring, ensuring greater efficacy and safety in the use of probiotics as a therapeutic strategy.
The connection between the gut microbiota and mental health has also emerged as a relevant aspect, especially in relation to depression in the elderly [74]. The bidirectional relationship between the gut and the brain suggests that modulation of the microbiota may directly influence depressive symptoms, regulating the production of neurotransmitters and reducing inflammatory markers. This opens new perspectives for the use of probiotics as adjuvants in the treatment of geriatric depression, which may represent a complementary approach to conventional therapies.
CONCLUSION
The findings of this review provide consistent evidence that probiotic supplementation may play a relevant role in improving cognitive and metabolic outcomes in older adults with chronic conditions, including diabetes mellitus, cardiovascular diseases, Alzheimer’s, Parkinson’s and depression. Modulation of the intestinal microbiota has emerged as a central factor in the regulation of inflammatory processes, energy metabolism and neurotransmission, suggesting that probiotics may act through multifactorial mechanisms to promote health benefits. However, despite the promising evidence, the high heterogeneity among the included studies imposes limitations on the interpretation of the results, highlighting the need for greater standardization in interventions, including the choice of probiotic strains, the optimal dose and the duration of treatment. Furthermore, the presence of publication bias and differences in the population studied reinforce the importance of conducting robust randomized clinical trials, with larger sample sizes and rigorous methodological criteria, to validate and expand the findings reported in this review.