INTRODUCTION
Guided Bone Regeneration (GBR) was introduced as a therapeutic modality that seeks the neoformation of resorbed bone tissue through the use of membranes1. The concept of GBR was established based on the principle of guided tissue regeneration, where certain tissues regenerate when cells with this capacity populate the defect during healing2.
The function of the membrane is to act as a physical barrier that prevents the migration of epithelial cells and connective tissue into the bone defect, while allowing osteogenic cells to stimulate bone tissue formation. Thus, many biodegradable and non-biodegradable materials have been used in the manufacture of regenerative membranes. Regardless of the type of material, regenerative membranes must exhibit biocompatibility (regulated in Brazil through NBR ISO 10993), integration with host tissues, clinical manageability, and the ability to maintain space3.
Non-biodegradable membranes are frequently used for periodontal and bone regeneration4, although they require a second surgical procedure for removal. On the other hand, biodegradable materials, although absorbable by the body and not requiring another intervention, have limitations in maintaining the physical barrier due to the membrane’s gradual degradation.
One type of membrane used in GBR (Guided Bone Regeneration) is the G21 Polyamide Membrane, a non-biodegradable material used in bone regeneration and the stabilization of dental implants. Its characteristics include impermeability and biocompatibility, which prevent microbial growth and the absorption of food residues, thus supporting Guided Bone Regeneration techniques. To ensure the microbiological safety of the product, it is necessary to determine the average microbial load according to the methods described in ISO 11737-1 (Sterilization of medical devices – Microbiological methods – Part 1: Determination of the population of microorganisms on product). This analysis must be carried out for each batch and for all samples, resulting in the total average microbial load. This control is conducted internally by the company Materplastic through a Bioburden study, ensuring the microbiological quality of the material5.
METHODS
Study Design
The study is characterized as a prospective clinical case report, focusing on the evaluation of the efficacy and safety of the G21 dental regenerative membrane (polyamide-based) in Guided Bone Regeneration (GBR) techniques.
The study phases include: Single intervention - Implantation of the G21 membrane after immediate extraction of element 16 (first upper right molar); Sequential monitoring - Clinical and imaging evaluations (radiographs and computed tomography) in the preoperative phase (analysis of the bone defect), immediate postoperative phase (15 days: membrane removal and analysis of granulation tissue) and late postoperative phase (80 days: evaluation of bone formation and viability for implant installation); and Internal comparison - Analysis of bone regeneration without the use of grafts or bone substitutes, using only the blood clot stabilized by the physical barrier (membrane).
Study Location and Period
Performed at a dental clinic in São Paulo, Brazil, under the responsibility of dentist Dr. Renan Nogueira da Costa (CRO-SP 116445).
The follow-up period began with the primary intervention on May 10, 2023, when tooth #16 was extracted, followed by the immediate implantation of the G21 regenerative membrane. The second stage occurred on May 25, 2023, corresponding to the 15th postoperative day, when the membrane was removed and the granulation tissue was assessed. Finally, on July 29, 2023, equivalent to 80 days after the initial surgery, the final evaluation was performed using clinical and imaging examinations, thus concluding the case’s monitoring cycle.
Study Population and Eligibility Criteria
A single participant was a 43-year-old male, classified as having normal system function. The patient had a formal indication for extraction of the upper right first molar (tooth 16) due to the presence of an extensive periradicular lesion and the impossibility of endodontic retreatment. The inclusion criteria were the need to preserve the alveolar ridge immediately after extraction and the absence of active infectious processes in the surgical region. In addition to the absence of known hypersensitivity to the components of polyamide (as per the manufacturer’s contraindications), a history of systemic comorbidities that compromise the regenerative cascade—such as uncontrolled diabetes mellitus or states of immunosuppression—as well as active smoking habits or the use of anti-angiogenic drugs, due to their potential to interfere with tissue repair processes.
Data Analysis
The data were evaluated using descriptive methods, focusing on the direct observation of clinical and imaging results in relation to granulation tissue and new bone formation.
Assessment of bone formation, measured by computed tomography (bone thickness, ridge height, and density); Clot integrity, in relation to the stability of the surgical site and absence of early membrane exposure; Complications, with records of inflammation, infection, or rejection, by means of panoramic radiographs, parasagittal and multiplanar tomographic sections, and 3D reconstructions.
Clinical success criteria were painless membrane removal within 15 days and absence of inflammatory signs. Functional success criteria included the feasibility of implant placement within 80 days (bone drilling without complications).
Ethical and Legal Aspects of Research
The polyamide film is manufactured by an approved supplier and is used exclusively by Materplastic for the production of dental regenerative membranes, having undergone rigorous biocompatibility testing. The biomaterial has been submitted for notification to ANVISA (National Health Surveillance Agency), as it meets the biocompatibility requirements of ISO 10993. It is submitted through a technical dossier with risk analysis, mitigating any risk of incompatibility with humans.
According to NBR ISO 10993-1, the Biocompatibility studies were completed, and the product was classified as low risk (Class II). Therefore, there is no need for Clinical studies, according to art. 3 of RDC of February 10, 2015 – Regularization of clinical trials with medical devices in Brazil.
Therefore, the reports are sufficient to prove Biocompatibility and will be attached to this report proving the veracity of the facts:
According to the Analysis report RL2232502CT, carried out by the certified laboratory ALS, the Dental Regenerative Membrane - G21 sample did not present a toxic effect on the L-929 cell line, therefore it was not considered cytotoxic.
According to the Analysis report RL2232500IMO, carried out by the certified laboratory, under the test conditions, the dental regenerative membrane sample-G21 was considered non-irritating to the oral mucosa of Syrian hamsters ( Mesocricetus auratus ).
According to the Analysis Report RL2232501LLNA, performed by the ALS-certified laboratory, under the test conditions, the Dental Regenerative Membrane - G21 sample presented a borderline positive result (SI between 1.6 and 1.9). Therefore, additional information was considered, as evidence of systemic toxicity. The animals did not show clinical signs of systemic toxicity.
The procedure strictly followed the ethical and legal precepts applicable to research involving human subjects and dental practice. Based on the Dental Code of Ethics and CNS Resolution 466/2012, the procedure ensured an assessment of risks and benefits. The biomaterial (polyamide G21) was used after satisfactory completion of biocompatibility studies, an essential step to ensure the minimum safety required before human exposure. The patient, duly informed of the nature of the new biomaterial and the procedure, provided free and informed consent, thus respecting the principle of autonomy. The technical execution was performed exclusively by a qualified dental surgeon (Dr. Renan Nogueira da Costa, CRO-SP 116445), in accordance with the legal attributions of the profession. The entire process ensured respect for the participant’s dignity and compliance with good clinical practice guidelines.
CASE PRESENTATION
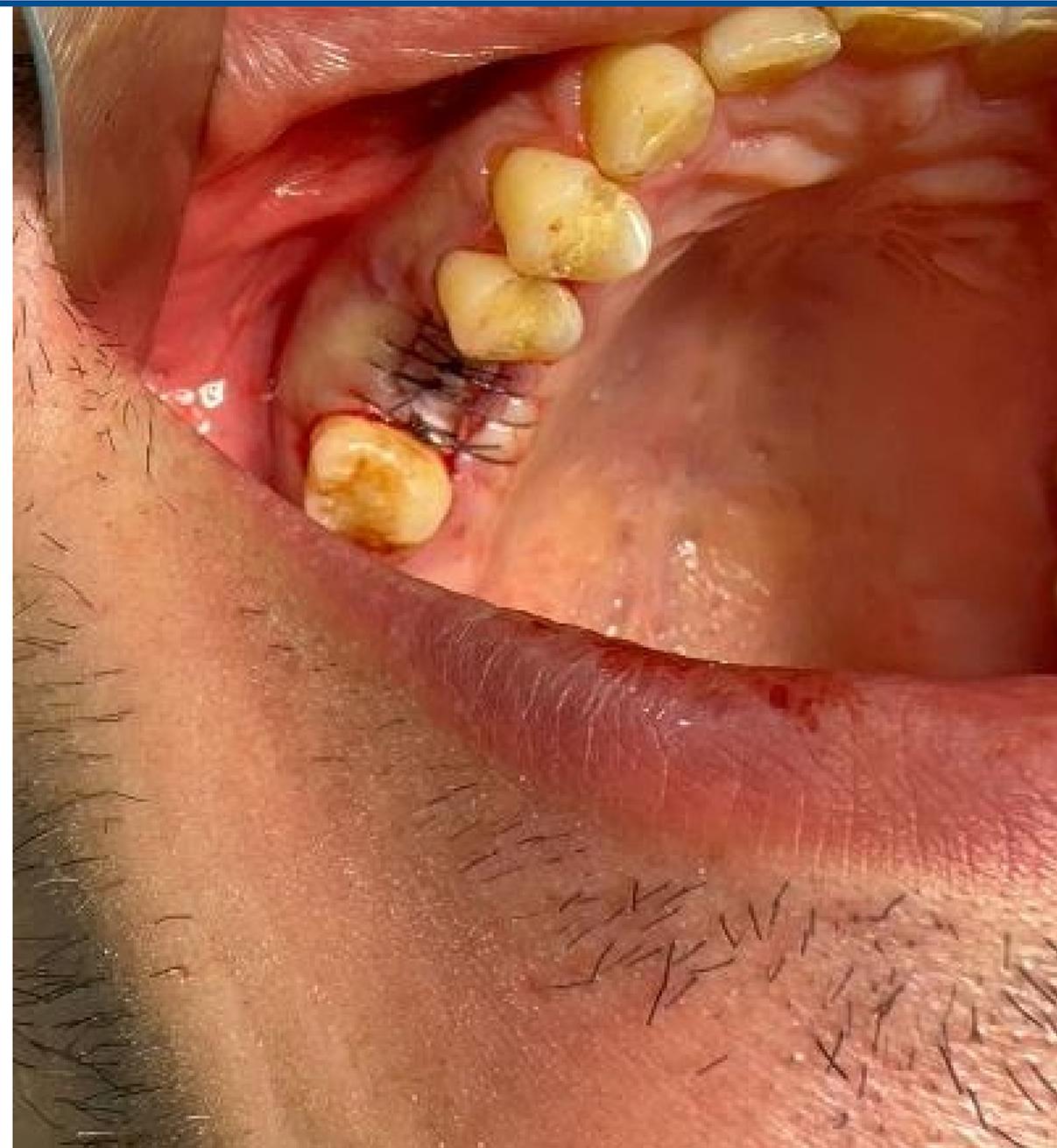
The 43-year-old male patient, of color He was white and classified as normosystem, living in the Brazilian state of São Paulo. He presented with intense pain when chewing and an increase in volume in the vestibular region of the upper right first molar (tooth 16). The tooth in question had previously undergone endodontic treatment and reconstruction with prefabricated posts cemented into the canals with resin cement. The tooth still presented an extensive apical lesion, and endodontic retreatment was not foreseeable. Therefore, we opted for extraction of the tooth (on May 10, 2023) and alveolar preservation with the G21 membrane.
In cases of alveolar preservation with injury, the use of bone grafts is not indicated due to the possibility of contamination of the biomaterial at the site, therefore the use of the G21 dental regenerative membrane is indicated, with the aim of preserving the site to favor the formation and stability of the blood clot through the ROG technique - Guided Bone Regeneration.
Application of the G21 Dental Regenerative Membrane
After completion of the biocompatibility studies, followed by the extraction of element 16, the G21 polyamide dental regenerative membrane was implanted in the patient and kept in place for 15 days.
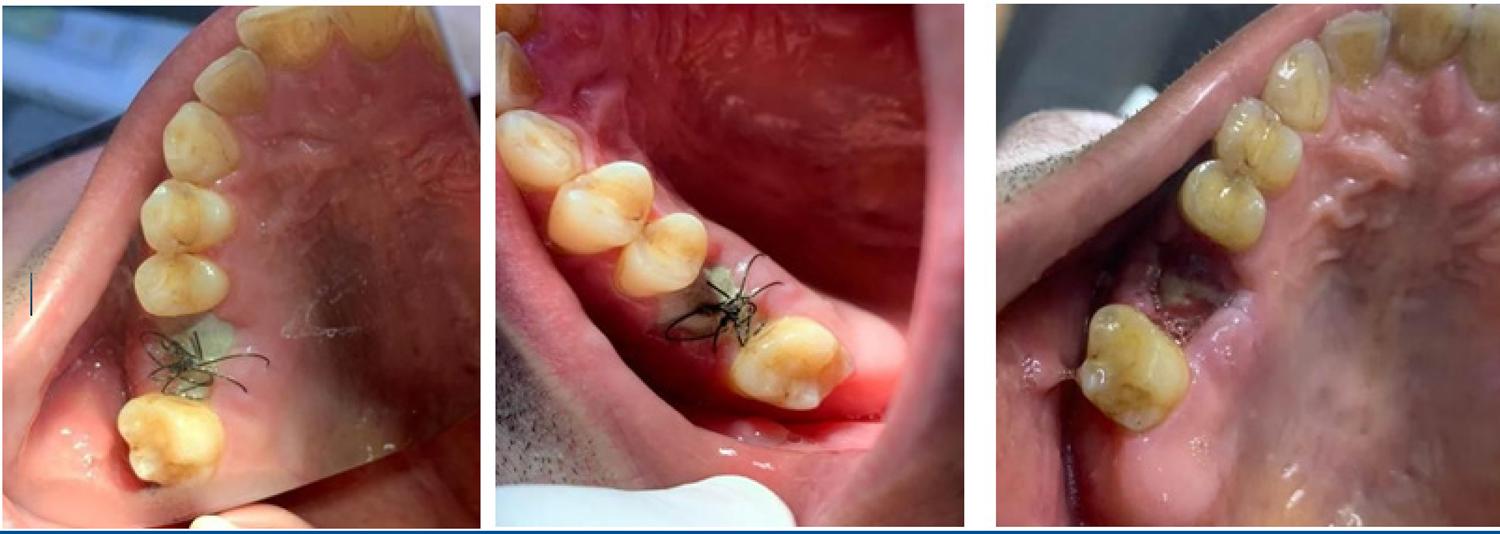
The G21 polyamide membrane was used as shown in Figure 3, offering high predictability of bone regeneration. The polyamide film was constructed with a smooth top, mitigating the risk of bacterial contamination, and is impermeable to solutions and food in the oral environment.
The patient was monitored during the membrane implantation period, and no inflammatory reactions were observed that would justify its removal.
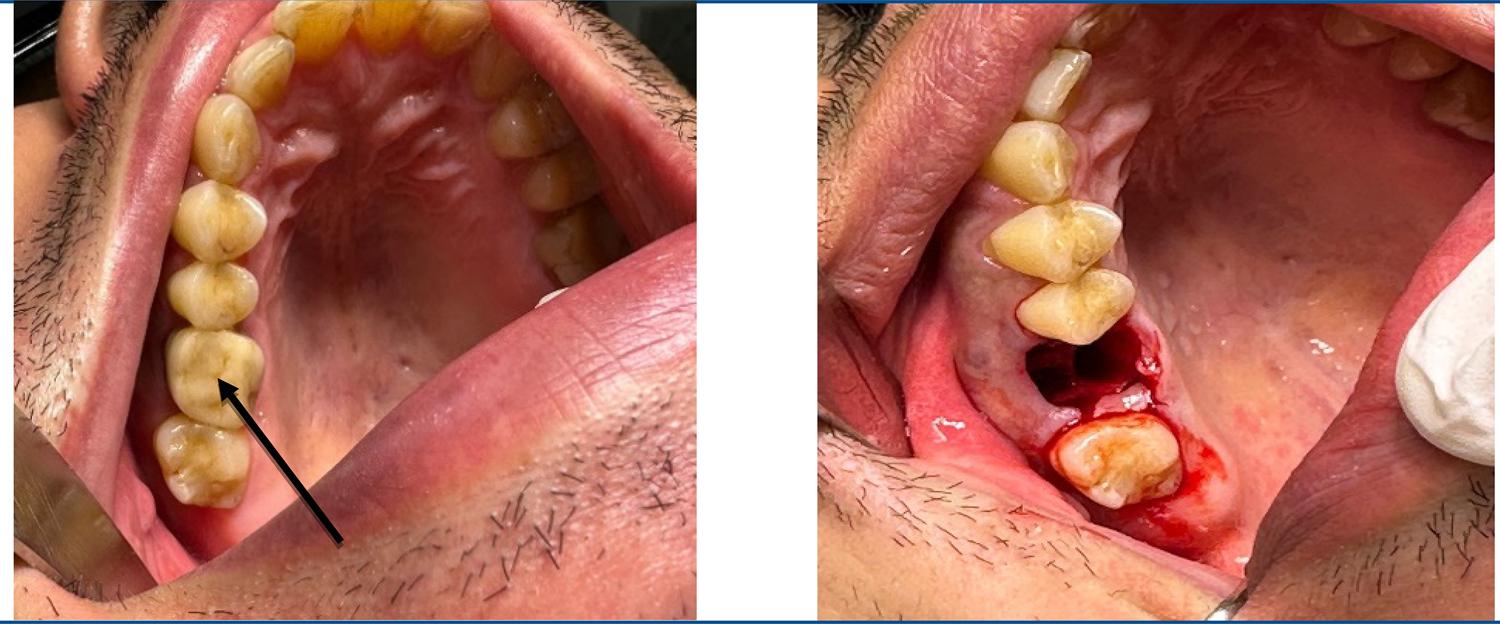
Removal of the G21 dental regenerative membrane, after 15 days
After the seventh day, the clot was replaced with granulation tissue, allowing the regenerative membrane to be removed. However, we opted to wait 15 days for this tissue to mature.
The dental regenerative membrane was removed 15 days after the initial implantation, in a simple and painless way that did not require further surgery or local anesthesia. The suture thread was removed and the membrane was removed with the aid of sterile tweezers.
Patient after 80 days of initial surgery
After the osteogenesis period was completed, the patient returned to the office, and the results were satisfactory, the expected formation occurred and we began the procedure for drilling the hole for the dental implant (Figures 5 and 6).
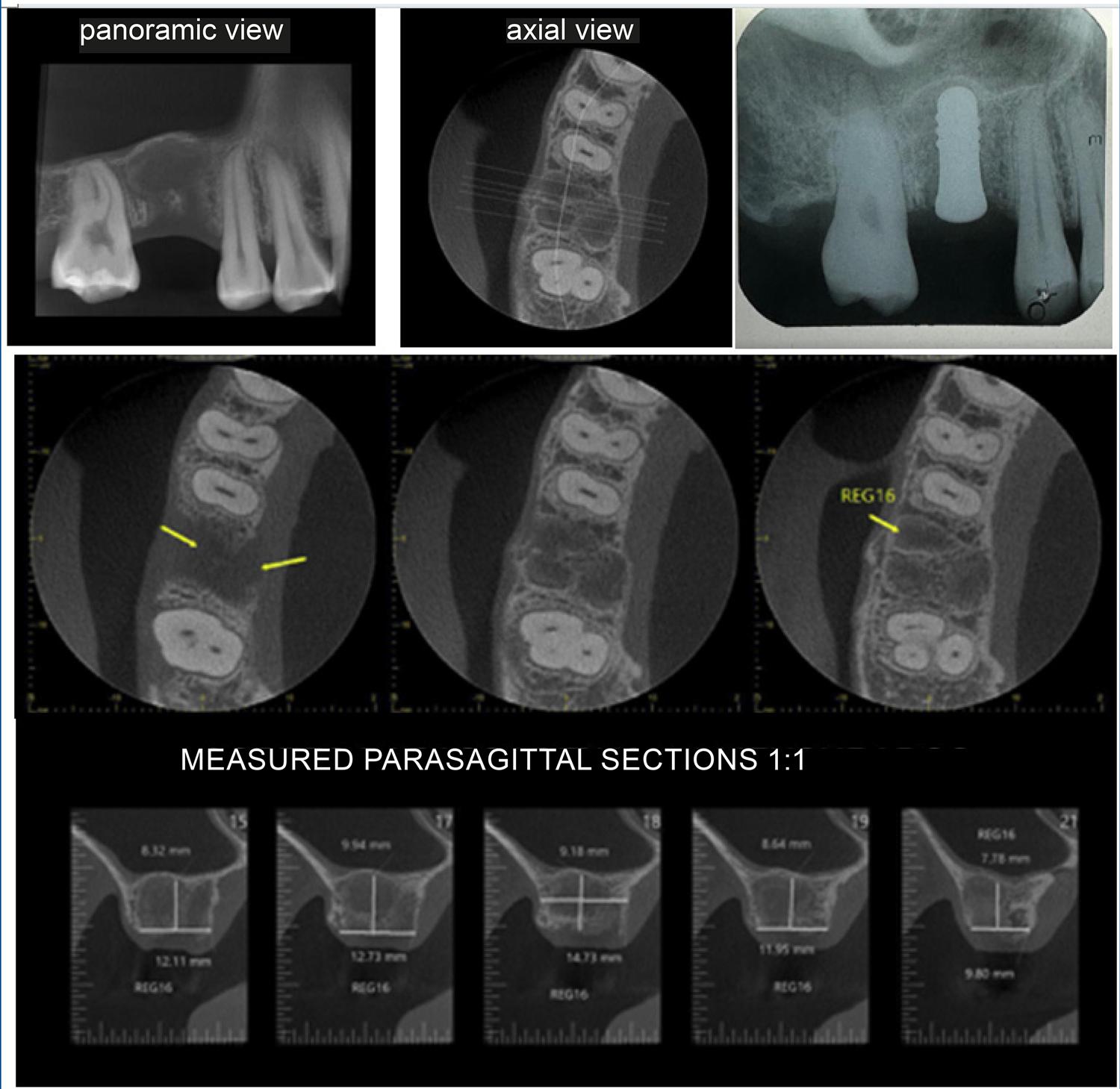
Figures 6 : radiographic aspects after extraction and 80-day evolution, in panoramic, axial and parasagittal views.
Analysis of the imaging results demonstrates the formation of newly formed bone tissue, indicating that the goal of Guided Bone Regeneration (GBR) was successfully achieved with the use of the new polyamide membrane, even in the absence of grafts or bone substitutes. The G21 regenerative dental membrane demonstrated satisfactory performance in relation to the fundamental criteria for this type of approach, including biocompatibility, space maintenance, cell exclusion, structural integrity, and appropriate clinical management. Furthermore, its application effectively contributed to promoting osteogenesis at the treated site.
DISCUSSION
The G21 dental regenerative membrane, after meeting the regulatory biocompatibility requirements, demonstrates clinical applicability in post-extraction cases, alveolar wall defects, immediate implants, small bone fenestrations and bone expansion procedures to increase thickness when bone marrow is present between the vestibular/palatal or vestibular/lingual cortices.
As established in the literature 6 , the blood clot is a fundamental element in the healing of the residual alveolar ridge, requiring preservation at the extraction site. However, studies show progressive alveolar resorption in short- and long-term analyses, making the maintenance of alveolar architecture critical for adequate functional and aesthetic rehabilitation7-8.
The Guided Bone Regeneration (GBR) technique employed allowed physiological preservation of the ridge dimensions without the use of grafts or bone substitutes. The alveolus was filled exclusively with blood clot and protected by the physical barrier of the G21 membrane, with imaging documentation demonstrating the formation of granulation tissue as a precursor stage of osteogenesis. As reported in previous studies, the osteogenic process begins immediately through clot organization, with complete bone maturation occurring within 90 days postoperatively, a period during which the membrane effectively prevented microbial contamination even after exposure to the oral environment9.
Analysis of the polyamide’s properties indicated satisfactory biocompatibility, with no evidence of cytotoxic effects or adverse tissue reactions in peri-implant tissues, recognizing that no implantable material exhibits absolute inertia. Its optimized thickness provides adequate rigidity for maintaining the regenerative space, resisting masticatory compressive forces without structural collapse, a crucial factor for preserving newly formed bone volume.
The effectiveness of cell exclusion was confirmed by the absence of fibrous connective tissue formation by epithelial migration, while the porous microarchitecture allowed selective diffusion of biological fluids and bioactive mediators, creating a microenvironment favorable to angiogenesis and osteoprogenitor differentiation.
CONCLUSION
The material’s thickness proved ideal for clinical handling, allowing for precise cutting, shaping, and adjustment to the bone defect area without the need for secondary surgical removal—a distinct advantage over other membranes available on the Brazilian market. Additionally, the G21 membrane exhibited excellent biocompatibility, effectively preventing microbial contamination and maintaining its integrity because it is non-resorbable.
Its stability was essential for the success of guided bone regeneration, as demonstrated by the results presented. Although the study did not monitor the final osseointegration phase of the implants, it achieved its primary objective: to validate the efficacy and safety of the G21 regenerative membrane, which is suitable for introduction into the Brazilian dental market.

Figure 2 : Radiographic appearance of element 16, before extraction: parasagittal, axial, panoramic view (ab), multiplanar ( c ), and three-dimensional ( d ) sections.











 texto em
texto em